Timing of Maternal COVID-19 Vaccine and Antibody Concentrations in Infants Born Preterm
- PMID: 38241046
- PMCID: PMC10799259
- DOI: 10.1001/jamanetworkopen.2023.52387
Timing of Maternal COVID-19 Vaccine and Antibody Concentrations in Infants Born Preterm
Abstract
Importance: COVID-19 vaccine-derived antibodies in pregnant people may protect infants from severe infection in the first 6 months of life via transplacental antibody transfer. Few data exist on maternally derived SARS-CoV-2 antibodies in preterm compared with full-term infants in association with vaccination timing.
Objective: To compare SARS-CoV-2 anti-Spike (anti-S) antibody levels in preterm and full-term infants in the context of vaccine dose timing before delivery.
Design, setting, and participants: This prospective cohort study enrolled pregnant individuals and collected paired maternal and cord blood samples at delivery at the University of Washington between February 1, 2021, and January 31, 2023. Participants who had received at least 2 doses of a messenger RNA COVID-19 vaccine before delivery and did not have a history of prior COVID-19 infection or detectable anti-SARS-CoV-2 nucleocapsid antibodies were included.
Exposures: Timing of the last vaccine dose and preterm or full-term gestational age at delivery.
Main outcomes and measures: Paired maternal and cord samples were tested for anti-S antibody, and linear regression was used to evaluate associations between preterm delivery and anti-S antibody levels. Covariates included timing of last dose, number of doses, insurance status, and immunosuppressing medications.
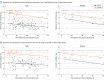
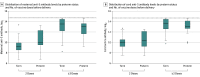
Results: A total of 220 participants (median [IQR] age, 34 [32-37] years; 212 [96.4%] female) with 36 preterm and 184 full-term deliveries were studied. Before delivery, 121 persons received 2 vaccine doses and 99 persons received 3 or more vaccine doses. The geometric mean concentration of maternal anti-S antibodies was 674 (95% CI, 577-787) after 2 doses and 8159 (95% CI, 6636-10 032) after 3 or more doses (P < .001). The cord anti-S antibody geometric mean concentration was 1000 (95% CI, 874-1144) after 2 doses and 9992 (95% CI, 8381-11 914) after 3 or more doses (P < .001). After adjustment for vaccine timing and number of doses before delivery, no association was found between preterm delivery and cord anti-S antibody levels (β = 0.44; 95% CI, -0.06 to 0.94).
Conclusions and relevance: In this prospective cohort study of pregnant individuals with preterm and full-term deliveries, receipt of 3 or more compared with 2 doses of COVID-19 vaccine before delivery resulted in 10-fold higher cord anti-S antibody levels. Maternal antibody concentration appeared more important than delivery gestational age in determining cord anti-S antibody levels. The number of doses and timing considerations for COVID-19 vaccine in pregnancy should include individuals at risk for preterm delivery.
Conflict of interest statement
Figures
References
-
- Delahoy MJ, Whitaker M, O’Halloran A, et al. ; COVID-NET Surveillance Team . Characteristics and maternal and birth outcomes of hospitalized pregnant women with laboratory-confirmed COVID-19—COVID-NET, 13 states, March 1-August 22, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(38):1347-1354. doi:10.15585/mmwr.mm6938e1 - DOI - PMC - PubMed
-
- Zambrano LD, Ellington S, Strid P, et al. ; CDC COVID-19 Response Pregnancy and Infant Linked Outcomes Team . Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(44):1641-1647. doi:10.15585/mmwr.mm6944e3 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

