Dual electrical stimulation at spinal-muscular interface reconstructs spinal sensorimotor circuits after spinal cord injury
- PMID: 38242904
- PMCID: PMC10799086
- DOI: 10.1038/s41467-024-44898-9
Dual electrical stimulation at spinal-muscular interface reconstructs spinal sensorimotor circuits after spinal cord injury
Abstract
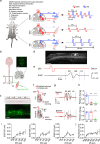
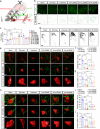
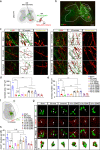
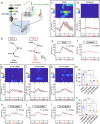
The neural signals produced by varying electrical stimulation parameters lead to characteristic neural circuit responses. However, the characteristics of neural circuits reconstructed by electrical signals remain poorly understood, which greatly limits the application of such electrical neuromodulation techniques for the treatment of spinal cord injury. Here, we develop a dual electrical stimulation system that combines epidural electrical and muscle stimulation to mimic feedforward and feedback electrical signals in spinal sensorimotor circuits. We demonstrate that a stimulus frequency of 10-20 Hz under dual stimulation conditions is required for structural and functional reconstruction of spinal sensorimotor circuits, which not only activates genes associated with axonal regeneration of motoneurons, but also improves the excitability of spinal neurons. Overall, the results provide insights into neural signal decoding during spinal sensorimotor circuit reconstruction, suggesting that the combination of epidural electrical and muscle stimulation is a promising method for the treatment of spinal cord injury.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Effects of tail nerve electrical stimulation on the activation and plasticity of the lumbar locomotor circuits and the prevention of skeletal muscle atrophy after spinal cord transection in rats.CNS Neurosci Ther. 2024 Mar;30(3):e14445. doi: 10.1111/cns.14445. Epub 2023 Sep 26. CNS Neurosci Ther. 2024. PMID: 37752787 Free PMC article.
-
A computational model for epidural electrical stimulation of spinal sensorimotor circuits.J Neurosci. 2013 Dec 4;33(49):19326-40. doi: 10.1523/JNEUROSCI.1688-13.2013. J Neurosci. 2013. PMID: 24305828 Free PMC article.
-
Epidural electrical stimulation of the cervical spinal cord opposes opioid-induced respiratory depression.J Physiol. 2022 Jun;600(12):2973-2999. doi: 10.1113/JP282664. Epub 2022 May 31. J Physiol. 2022. PMID: 35639046
-
Emergence of Epidural Electrical Stimulation to Facilitate Sensorimotor Network Functionality After Spinal Cord Injury.Neuromodulation. 2019 Apr;22(3):244-252. doi: 10.1111/ner.12938. Epub 2019 Mar 6. Neuromodulation. 2019. PMID: 30840354 Review.
-
Probing the Human Spinal Locomotor Circuits by Phasic Step-Induced Feedback and by Tonic Electrical and Pharmacological Neuromodulation.Curr Pharm Des. 2017;23(12):1805-1820. doi: 10.2174/1381612822666161214144655. Curr Pharm Des. 2017. PMID: 27981912 Review.
Cited by
-
Prediction of isometric forces from combined epidural spinal cord and neuromuscular electrical stimulation in the rat lower limb.Sci Rep. 2024 Jul 9;14(1):15871. doi: 10.1038/s41598-024-66773-9. Sci Rep. 2024. PMID: 38982137 Free PMC article.
-
Spatiotemporal spinal cord stimulation with real-time triggering exoskeleton restores walking capability: a case report.Ann Clin Transl Neurol. 2025 Mar;12(3):659-665. doi: 10.1002/acn3.52281. Epub 2024 Dec 15. Ann Clin Transl Neurol. 2025. PMID: 39675019 Free PMC article.
-
Chronic motoneuronal activation enhanced axonal regeneration and functional recovery after brachial plexus injury.J Orthop Translat. 2025 Apr 2;52:40-54. doi: 10.1016/j.jot.2025.02.007. eCollection 2025 May. J Orthop Translat. 2025. PMID: 40235635 Free PMC article.
-
Chronic Intermittent Hypoxia-Induced Neural Injury: Pathophysiology, Neurodegenerative Implications, and Therapeutic Insights.CNS Neurosci Ther. 2025 Apr;31(4):e70384. doi: 10.1111/cns.70384. CNS Neurosci Ther. 2025. PMID: 40260643 Free PMC article. Review.
-
Effects of Teriparatide and Alendronate on Functional Recovery from Spinal Cord Injury and Postinjury Bone Loss.Biomedicines. 2025 Feb 3;13(2):342. doi: 10.3390/biomedicines13020342. Biomedicines. 2025. PMID: 40002755 Free PMC article.
References
MeSH terms
Grants and funding
- 82171376/National Natural Science Foundation of China (National Science Foundation of China)
- 81971164/National Natural Science Foundation of China (National Science Foundation of China)
- 81771330/National Natural Science Foundation of China (National Science Foundation of China)
- 81330026/National Natural Science Foundation of China (National Science Foundation of China)
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

