Discovery of a peripheral 5HT2A antagonist as a clinical candidate for metabolic dysfunction-associated steatohepatitis
- PMID: 38245505
- PMCID: PMC10799935
- DOI: 10.1038/s41467-024-44874-3
Discovery of a peripheral 5HT2A antagonist as a clinical candidate for metabolic dysfunction-associated steatohepatitis
Abstract
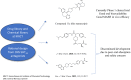
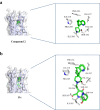
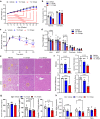
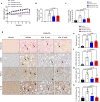
Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) is currently the leading cause of chronic liver disease worldwide. Metabolic Dysfunction-Associated Steatohepatitis (MASH), an advanced form of MASLD, can progress to liver fibrosis, cirrhosis, and hepatocellular carcinoma. Based on recent findings by our team that liver 5HT2A knockout male mice suppressed steatosis and reduced fibrosis-related gene expression, we developed a peripheral 5HT2A antagonist, compound 11c for MASH. It shows good in vitro activity, stability, and in vivo pharmacokinetics (PK) in rats and dogs. Compound 11c also shows good in vivo efficacy in a diet-induced obesity (DIO) male mice model and in a choline-deficient, L-amino acid-defined, high-fat diet (CDAHFD) male mice model, effectively improving histologic features of MASH and fibrosis. According to the tissue distribution study using [14C]-labeled 11c, the compound was determined to be a peripheral 5HT2A antagonist. Collectively, first-in-class compound 11c shows promise as a therapeutic agent for the treatment of MASLD and MASH.
© 2024. The Author(s).
Conflict of interest statement
A.J.H., H.S.P., S.H.P., M.H.K. and W.M.L. have filed a patent application encompassing aspects of this work (US 2023/0002349 A1 and WO 2021/086133 A1). The remaining authors declare no competing interests.
Figures
References
-
- Rinella ME, et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;29:101133. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

