Engineering strategies for enhanced heterologous protein production by Saccharomyces cerevisiae
- PMID: 38247006
- PMCID: PMC10801990
- DOI: 10.1186/s12934-024-02299-z
Engineering strategies for enhanced heterologous protein production by Saccharomyces cerevisiae
Abstract
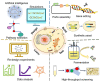
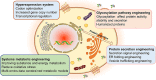
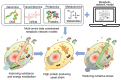
Microbial proteins are promising substitutes for animal- and plant-based proteins. S. cerevisiae, a generally recognized as safe (GRAS) microorganism, has been frequently employed to generate heterologous proteins. However, constructing a universal yeast chassis for efficient protein production is still a challenge due to the varying properties of different proteins. With progress in synthetic biology, a multitude of molecular biology tools and metabolic engineering strategies have been employed to alleviate these issues. This review first analyses the advantages of protein production by S. cerevisiae. The most recent advances in improving heterologous protein yield are summarized and discussed in terms of protein hyperexpression systems, protein secretion engineering, glycosylation pathway engineering and systems metabolic engineering. Furthermore, the prospects for efficient and sustainable heterologous protein production by S. cerevisiae are also provided.
Keywords: Expression system; Protein production; Saccharomyces cerevisiae; Secretion engineering; Systems metabolic engineering.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Multidimensional strategies for efficient heterologous protein expression in Aspergillus niger.Biotechnol Adv. 2025 Aug 20;84:108690. doi: 10.1016/j.biotechadv.2025.108690. Online ahead of print. Biotechnol Adv. 2025. PMID: 40845950 Review.
-
Metabolic engineering and synthetic biology for isoprenoid production in Escherichia coli and Saccharomyces cerevisiae.Appl Microbiol Biotechnol. 2021 Jan;105(2):457-475. doi: 10.1007/s00253-020-11040-w. Epub 2021 Jan 4. Appl Microbiol Biotechnol. 2021. PMID: 33394155 Review.
-
Recent advances in construction and regulation of yeast cell factories.World J Microbiol Biotechnol. 2022 Feb 17;38(4):57. doi: 10.1007/s11274-022-03241-4. World J Microbiol Biotechnol. 2022. PMID: 35174424 Review.
-
[Metabolic engineering tools for Saccharomyces cerevisiae].Sheng Wu Gong Cheng Xue Bao. 2021 May 25;37(5):1578-1602. doi: 10.13345/j.cjb.200690. Sheng Wu Gong Cheng Xue Bao. 2021. PMID: 34085444 Review. Chinese.
-
Microbial chassis engineering drives heterologous production of complex secondary metabolites.Biotechnol Adv. 2022 Oct;59:107966. doi: 10.1016/j.biotechadv.2022.107966. Epub 2022 Apr 26. Biotechnol Adv. 2022. PMID: 35487394 Review.
Cited by
-
Advancing cellulose utilization and engineering consolidated bioprocessing yeasts: current state and perspectives.Appl Microbiol Biotechnol. 2025 Feb 13;109(1):43. doi: 10.1007/s00253-025-13426-0. Appl Microbiol Biotechnol. 2025. PMID: 39939397 Free PMC article. Review.
-
Multiplex genetic manipulations in Clostridium butyricum and Clostridium sporogenes to secrete recombinant antigen proteins for oral-spore vaccination.Microb Cell Fact. 2024 Apr 24;23(1):119. doi: 10.1186/s12934-024-02389-y. Microb Cell Fact. 2024. PMID: 38659027 Free PMC article.
-
A single-vector CRISPR/Cas9 system for genome editing and heterologous enzyme secretion in Saccharomyces cerevisiae: a case study on pectate lyase for coffee mucilage removal.Biotechnol Lett. 2025 Jul 19;47(4):78. doi: 10.1007/s10529-025-03621-4. Biotechnol Lett. 2025. PMID: 40684030
-
Synthetic Biology in Natural Product Biosynthesis.Chem Rev. 2025 Apr 9;125(7):3814-3931. doi: 10.1021/acs.chemrev.4c00567. Epub 2025 Mar 21. Chem Rev. 2025. PMID: 40116601 Review.
-
Facile expression of proteins with desired N-terminal amino acid via an engineered cysteine protease domain.Commun Biol. 2025 Aug 6;8(1):1165. doi: 10.1038/s42003-025-08614-7. Commun Biol. 2025. PMID: 40770049 Free PMC article.
References
-
- Arnthong J, Ponjarat J, Bussadee P, Deenarn P, Prommana P, Phienluphon A, Charoensri S, Champreda V, Zhao X-Q, Suwannarangsee S. Enhanced surface display efficiency of β-glucosidase in Saccharomyces cerevisiae by disruption of cell wall protein-encoding genes YGP1 and CWP2. Biochem Eng J. 2022;179.
-
- Ren S, Hu P, Jia J, Ni J, Jiang T, Yang H, Bai J, Tian C, Chen L, Huang Q et al. Engineering of Saccharomyces cerevisiae for sensing sweetness. Biochem Eng J. 2022;177.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

