The Detection of Primary Sclerosing Cholangitis Using Volatile Metabolites in Fecal Headspace and Exhaled Breath
- PMID: 38248826
- PMCID: PMC10819709
- DOI: 10.3390/metabo14010023
The Detection of Primary Sclerosing Cholangitis Using Volatile Metabolites in Fecal Headspace and Exhaled Breath
Abstract
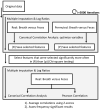
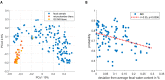
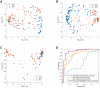
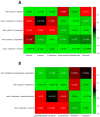
Up to 5% of inflammatory bowel disease patients may at some point develop primary sclerosing cholangitis (PSC). PSC is a rare liver disease that ultimately results in liver damage, cirrhosis and liver failure. It typically remains subclinical until irreversible damage has been inflicted. Hence, it is crucial to screen IBD patients for PSC, but its early detection is challenging, and the disease's etiology is not well understood. This current study aimed at the early detection of PSC in an IBD population using Volatile Organic Compounds in fecal headspace and exhaled breath. To this aim, fecal material and exhaled breath were collected from 73 patients (n = 16 PSC/IBD; n = 8 PSC; n = 49 IBD), and their volatile profile were analyzed using Gas Chromatography-Mass Spectrometry. Using the most discriminatory features, PSC detection resulted in areas under the ROC curve (AUCs) of 0.83 and 0.84 based on fecal headspace and exhaled breath, respectively. Upon data fusion, the predictive performance increased to AUC 0.92. The observed features in the fecal headspace relate to detrimental microbial dysbiosis and exogenous exposure. Future research should aim for the early detection of PSC in a prospective study design.
Keywords: early detection; exhaled breath; fecal headspace; inflammatory bowel disease; liver disease; metabolomics; primary sclerosing cholangitis; volatile organic compounds.
Conflict of interest statement
The authors declare no conflicts of interest related to the current study. AS is an assistant professor at Maastricht University and is an advisor at Owlstone Medical (Cambridge, UK), a breath-based medical company aiming for the non-invasive detection of diseases.
Figures


Similar articles
-
The potential of volatile organic compounds to diagnose primary sclerosing cholangitis.JHEP Rep. 2024 Apr 27;6(8):101103. doi: 10.1016/j.jhepr.2024.101103. eCollection 2024 Aug. JHEP Rep. 2024. PMID: 39131082 Free PMC article.
-
Alterations of the salivary and fecal microbiome in patients with primary sclerosing cholangitis.Hepatol Int. 2021 Feb;15(1):191-201. doi: 10.1007/s12072-020-10089-z. Epub 2020 Sep 19. Hepatol Int. 2021. PMID: 32949377
-
Symptoms Do Not Correlate With Findings From Colonoscopy in Children With Inflammatory Bowel Disease and Primary Sclerosing Cholangitis.Clin Gastroenterol Hepatol. 2018 Jul;16(7):1098-1105.e1. doi: 10.1016/j.cgh.2018.01.020. Epub 2018 Mar 24. Clin Gastroenterol Hepatol. 2018. PMID: 29378308
-
Gut microbiome in primary sclerosing cholangitis: A review.World J Gastroenterol. 2020 Jun 7;26(21):2768-2780. doi: 10.3748/wjg.v26.i21.2768. World J Gastroenterol. 2020. PMID: 32550753 Free PMC article. Review.
-
Distinctive inflammatory bowel disease phenotype in primary sclerosing cholangitis.World J Gastroenterol. 2015 Feb 14;21(6):1956-71. doi: 10.3748/wjg.v21.i6.1956. World J Gastroenterol. 2015. PMID: 25684965 Free PMC article. Review.
Cited by
-
Progress and challenges of developing volatile metabolites from exhaled breath as a biomarker platform.Metabolomics. 2024 Jul 8;20(4):72. doi: 10.1007/s11306-024-02142-x. Metabolomics. 2024. PMID: 38977623 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

