Revisiting the two rhythm generators for respiration in lampreys
- PMID: 38250023
- PMCID: PMC10796688
- DOI: 10.3389/fnana.2023.1270535
Revisiting the two rhythm generators for respiration in lampreys
Abstract
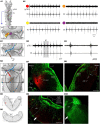
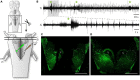
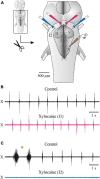
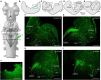
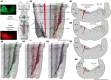
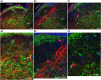
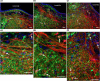
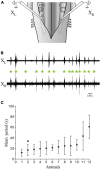
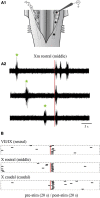
In lampreys, respiration consists of a fast and a slow rhythm. This study was aimed at characterizing both anatomically and physiologically the brainstem regions involved in generating the two rhythms. The fast rhythm generator has been located by us and others in the rostral hindbrain, rostro-lateral to the trigeminal motor nucleus. More recently, this was challenged by researchers reporting that the fast rhythm generator was located more rostrally and dorsomedially, in a region corresponding to the mesencephalic locomotor region. These contradictory observations made us re-examine the location of the fast rhythm generator using anatomical lesions and physiological recordings. We now confirm that the fast respiratory rhythm generator is in the rostro-lateral hindbrain as originally described. The slow rhythm generator has received less attention. Previous studies suggested that it was composed of bilateral, interconnected rhythm generating regions located in the caudal hindbrain, with ascending projections to the fast rhythm generator. We used anatomical and physiological approaches to locate neurons that could be part of this slow rhythm generator. Combinations of unilateral injections of anatomical tracers, one in the fast rhythm generator area and another in the lateral tegmentum of the caudal hindbrain, were performed to label candidate neurons on the non-injected side of the lateral tegmentum. We found a population of neurons extending from the facial to the caudal vagal motor nuclei, with no clear clustering in the cell distribution. We examined the effects of stimulating different portions of the labeled population on the respiratory activity. The rostro-caudal extent of the population was arbitrarily divided in three portions that were each stimulated electrically or chemically. Stimulation of either of the three sites triggered bursts of discharge characteristic of the slow rhythm, whereas inactivating any of them stopped the slow rhythm. Substance P injected locally in the lateral tegmentum accelerated the slow respiratory rhythm in a caudal hindbrain preparation. Our results show that the fast respiratory rhythm generator consists mostly of a population of neurons rostro-lateral to the trigeminal motor nucleus, whereas the slow rhythm generator is distributed in the lateral tegmentum of the caudal hindbrain.
Keywords: DAMGO; brainstem; electrophysiology; lamprey; neuroanatomy; respiration; respiratory generator; substance P.
Copyright © 2024 Missaghi, Le Gal, Mercier, Grover, Beauséjour, Chartré, Messihad, Auclair and Dubuc.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures






Similar articles
-
Respiratory rhythms generated in the lamprey rhombencephalon.Neuroscience. 2007 Aug 10;148(1):279-93. doi: 10.1016/j.neuroscience.2007.05.023. Epub 2007 Jul 6. Neuroscience. 2007. PMID: 17618060
-
Swimming rhythm generation in the caudal hindbrain of the lamprey.J Neurophysiol. 2018 May 1;119(5):1681-1692. doi: 10.1152/jn.00851.2017. Epub 2018 Jan 24. J Neurophysiol. 2018. PMID: 29364070 Free PMC article.
-
Breathing rhythm-generation mechanism in the adult lamprey (Lampetra japonica).Jpn J Physiol. 1984;34(2):319-35. doi: 10.2170/jjphysiol.34.319. Jpn J Physiol. 1984. PMID: 6471613
-
The neural control of respiration in lampreys.Respir Physiol Neurobiol. 2016 Dec;234:14-25. doi: 10.1016/j.resp.2016.08.007. Epub 2016 Aug 22. Respir Physiol Neurobiol. 2016. PMID: 27562521 Review.
-
Initiation of locomotion in lampreys.Brain Res Rev. 2008 Jan;57(1):172-82. doi: 10.1016/j.brainresrev.2007.07.016. Epub 2007 Aug 22. Brain Res Rev. 2008. PMID: 17916380 Review.
References
-
- Arata A., Tanaka I., Fujii M., Ezure K. (2010). “Active inspiratory-expiratory phase switching mechanism exists in the neonatal nucleus parabrachialis,” in New Frontiers in Respiratory Control Advances in Experimental Medicine and Biology, eds Homma I., Onimaru H., Fukuchi Y. (New York, NY: Springer; ), 135–138. 10.1007/978-1-4419-5692-7_27 - DOI - PubMed
LinkOut - more resources
Full Text Sources

