The predictive role of symptoms in COVID-19 diagnostic models: A longitudinal insight
- PMID: 38250791
- PMCID: PMC10945957
- DOI: 10.1017/S0950268824000037
The predictive role of symptoms in COVID-19 diagnostic models: A longitudinal insight
Abstract
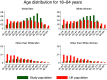
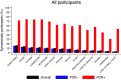
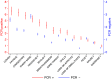
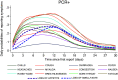
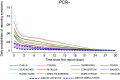
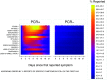
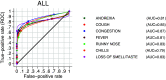
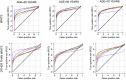
To investigate the symptoms of SARS-CoV-2 infection, their dynamics and their discriminatory power for the disease using longitudinally, prospectively collected information reported at the time of their occurrence. We have analysed data from a large phase 3 clinical UK COVID-19 vaccine trial. The alpha variant was the predominant strain. Participants were assessed for SARS-CoV-2 infection via nasal/throat PCR at recruitment, vaccination appointments, and when symptomatic. Statistical techniques were implemented to infer estimates representative of the UK population, accounting for multiple symptomatic episodes associated with one individual. An optimal diagnostic model for SARS-CoV-2 infection was derived. The 4-month prevalence of SARS-CoV-2 was 2.1%; increasing to 19.4% (16.0%-22.7%) in participants reporting loss of appetite and 31.9% (27.1%-36.8%) in those with anosmia/ageusia. The model identified anosmia and/or ageusia, fever, congestion, and cough to be significantly associated with SARS-CoV-2 infection. Symptoms' dynamics were vastly different in the two groups; after a slow start peaking later and lasting longer in PCR+ participants, whilst exhibiting a consistent decline in PCR- participants, with, on average, fewer than 3 days of symptoms reported. Anosmia/ageusia peaked late in confirmed SARS-CoV-2 infection (day 12), indicating a low discrimination power for early disease diagnosis.
Keywords: coronavirus; longitudinal data; symptoms dynamics.
Conflict of interest statement
C.A.C. reports receiving grant support, paid to her institution, from Novavax, Moderna, GSK. A.L.G. reports receiving grant support, paid to her institution, from Novavax and entered into a partnership with AstraZeneca for further development of ChAdOx1 nCoV-19. A.L.G. is named as an inventor on a patent covering the use of a particular promoter construct that is often used in vectored vaccines and is incorporated in the ChAdOx1 nCoV-19 vaccine and may benefit from royalty income paid to the University of Oxford from sales of this vaccine by AstraZeneca and its sublicensees under the university’s revenue sharing policy. P.T.H. reports receiving grant support, paid to his institution, from Novavax, Pfizer, Moderna, Valneva, Janssen, Astra Zeneca. I.C.S. declares receiving grant support, paid to her institution, from NIHR and Astra Zeneca. Other authors reported no competing interest.
Figures



References
-
- World Health Organisation (2023) WHO Coronavirus (COVID-19) Dashboard, WHO Coronavirus (COVID-19) Dashboard With Vaccination Data. Available at https://covid19.who.int/ (accessed 7 March 2023).
-
- Emanuel EJ, et al. (2020) Fair allocation of scarce medical resources in the time of COVID-19. The New England Journal of Medicine 382, 2049–2055. - PubMed
-
- National Health Service (2022) Main Symptoms of Coronavirus (COVID-19) – NHS. Available at https://www.nhs.uk/conditions/coronavirus-covid-19/symptoms/main-symptoms/ (accessed 30 March 2022).
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

