Mycobacterium tuberculosis Deficient in PdtaS Cytosolic Histidine Kinase Displays Attenuated Growth and Affords Protective Efficacy against Aerosol M. tuberculosis Infection in Mice
- PMID: 38250863
- PMCID: PMC10821411
- DOI: 10.3390/vaccines12010050
Mycobacterium tuberculosis Deficient in PdtaS Cytosolic Histidine Kinase Displays Attenuated Growth and Affords Protective Efficacy against Aerosol M. tuberculosis Infection in Mice
Abstract
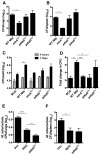
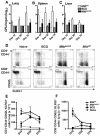
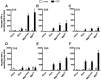
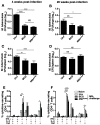
New control measures are urgently required to control tuberculosis (TB), as the current vaccine, Bacille Calmette-Guérin (BCG), has had a limited impact on disease spread. The identification of virulence mechanisms of Mycobacterium tuberculosis is an important strategy in vaccine design, as it permits the development of strains attenuated for growth that may have vaccine potential. In this report, we determined the role of the PdtaS response regulator in M. tuberculosis virulence and defined the vaccine potential of a pdtaS-deficient strain. Deletion of pdtaS (MtbΔpdtaS) resulted in reduced persistence of M. tuberculosis within mouse organs, which was equivalent to the persistence of the BCG vaccine in the lung and liver of infected mice. However, the generation of effector CD4+ and CD8+ T cells (CD44+CD62LloKLRG1+) was similar between wild-type M. tuberculosis and MtbΔpdtaS and greater than that elicited by BCG. Heightened immunity induced by MtbΔpdtaS compared to BCG was also observed by analysis of antigen-specific IFN-γ-secreting T cell responses induced by vaccination. MtbΔpdtaS displayed improved protection against aerosol M. tuberculosis compared to BCG, which was most apparent in the lung at 20 weeks post-infection. These results suggest that the deletion of the PdtaS response regulator warrants further appraisal as a tool to combat TB in humans.
Keywords: PdtaS; immune response; tuberculosis; vaccine; virulence.
Conflict of interest statement
The authors report no conflict of interest related to the content of this manuscript. The funders had no role in the design of the study, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
Immunogenicity and vaccine potential of clinical isolate Mycobacterium kansasii strain against Mycobacterium tuberculosis infection.Microbiol Spectr. 2024 Aug 6;12(8):e0081924. doi: 10.1128/spectrum.00819-24. Epub 2024 Jul 9. Microbiol Spectr. 2024. PMID: 38980025 Free PMC article.
-
Listeria-Vectored Multiantigenic Tuberculosis Vaccine Enhances Protective Immunity against Aerosol Challenge with Virulent Mycobacterium tuberculosis in BCG-Immunized C57BL/6 and BALB/c Mice.mBio. 2022 Jun 28;13(3):e0068722. doi: 10.1128/mbio.00687-22. Epub 2022 Jun 1. mBio. 2022. PMID: 35642945 Free PMC article.
-
Repeated Aerosolized-Boosting with Gamma-Irradiated Mycobacterium bovis BCG Confers Improved Pulmonary Protection against the Hypervirulent Mycobacterium tuberculosis Strain HN878 in Mice.PLoS One. 2015 Oct 28;10(10):e0141577. doi: 10.1371/journal.pone.0141577. eCollection 2015. PLoS One. 2015. PMID: 26509812 Free PMC article.
-
[Novel vaccines against M. tuberculosis].Kekkaku. 2006 Dec;81(12):745-51. Kekkaku. 2006. PMID: 17240920 Review. Japanese.
-
Is interferon-gamma the right marker for bacille Calmette-Guérin-induced immune protection? The missing link in our understanding of tuberculosis immunology.Clin Exp Immunol. 2012 Sep;169(3):213-9. doi: 10.1111/j.1365-2249.2012.04614.x. Clin Exp Immunol. 2012. PMID: 22861360 Free PMC article. Review.
References
-
- World Health Organisation . Global Tuberculosis Report 2023. WHO; Geneva, Switzerland: 2023.
-
- Spertini F., Audran R., Chakour R., Karoui O., Steiner-Monard V., Thierry A.C., Mayor C.E., Rettby N., Jaton K., Vallotton L., et al. Safety of human immunisation with a live-attenuated Mycobacterium tuberculosis vaccine: A randomised, double-blind, controlled phase I trial. Lancet Respir. Med. 2015;3:953–962. doi: 10.1016/S2213-2600(15)00435-X. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

