Antinociceptive Effects of Cannabichromene (CBC) in Mice: Insights from von Frey, Tail-Flick, Formalin, and Acetone Tests
- PMID: 38255191
- PMCID: PMC10813533
- DOI: 10.3390/biomedicines12010083
Antinociceptive Effects of Cannabichromene (CBC) in Mice: Insights from von Frey, Tail-Flick, Formalin, and Acetone Tests
Abstract
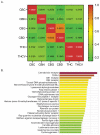
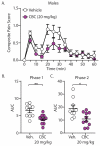
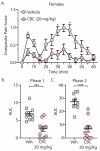
Cannabis sativa contains minor cannabinoids that have potential therapeutic value in pain management. However, detailed experimental evidence for the antinociceptive effects of many of these minor cannabinoids remains lacking. Here, we employed artificial intelligence (AI) to perform compound-protein interaction estimates with cannabichromene (CBC) and receptors involved in nociceptive signaling. Based on our findings, we investigated the antinociceptive properties of CBC in naïve or neuropathic C57BL/6 male and female mice using von Frey (mechanical allodynia), tail-flick (noxious radiant heat), formalin (acute and persistent inflammatory pain), and acetone (cold thermal) tests. For von Frey assessments, CBC dose (0-20 mg/kg, i.p.) and time (0-6 h) responses were measured in male and female neuropathic mice. For tail-flick, formalin, and acetone assays, CBC (20 mg/kg, i.p.) was administered to naïve male and female mice 1 h prior to testing. The results show that CBC (10 and 20 mg/kg, i.p.) significantly reduced mechanical allodynia in neuropathic male and female mice 1-2 h after treatment. Additionally, CBC treatment caused significant reductions in nociceptive behaviors in the tail-flick assay and in both phase 1 and phase 2 of the formalin test. Finally, we found a significant interaction in neuropathic male mice in the acetone test. In conclusion, our results suggest that CBC targets receptors involved in nociceptive signaling and imparts antinociceptive properties that may benefit males and females afflicted with diverse forms of acute or chronic/persistent pain.
Keywords: cannabichromene; cannabinoids; cannabis; inflammatory pain; neuropathic pain; pain.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
The antinociceptive effect of SNAP5114, a gamma-aminobutyric acid transporter-3 inhibitor, in rat experimental pain models.Anesth Analg. 2013 May;116(5):1162-1169. doi: 10.1213/ANE.0b013e318282dda7. Epub 2013 Mar 1. Anesth Analg. 2013. PMID: 23456665
-
Cannabigerol (CBG) attenuates mechanical hypersensitivity elicited by chemotherapy-induced peripheral neuropathy.Eur J Pain. 2022 Oct;26(9):1950-1966. doi: 10.1002/ejp.2016. Epub 2022 Aug 4. Eur J Pain. 2022. PMID: 35899583
-
The Antinociceptive Properties of the Corydalis yanhusuo Extract.PLoS One. 2016 Sep 13;11(9):e0162875. doi: 10.1371/journal.pone.0162875. eCollection 2016. PLoS One. 2016. PMID: 27622550 Free PMC article.
-
Differential activities of intrathecal MK-801 or morphine to alter responses to thermal and mechanical stimuli in normal or nerve-injured rats.Pain. 1997 May;71(1):57-64. doi: 10.1016/s0304-3959(97)03337-x. Pain. 1997. PMID: 9200174
-
The Potential of Cannabichromene (CBC) as a Therapeutic Agent.J Pharmacol Exp Ther. 2024 Oct 18;391(2):206-213. doi: 10.1124/jpet.124.002166. J Pharmacol Exp Ther. 2024. PMID: 38777605 Free PMC article. Review.
Cited by
-
Cannabis-Based Phytocannabinoids: Overview, Mechanism of Action, Therapeutic Application, Production, and Affecting Environmental Factors.Int J Mol Sci. 2024 Oct 19;25(20):11258. doi: 10.3390/ijms252011258. Int J Mol Sci. 2024. PMID: 39457041 Free PMC article. Review.
-
CANDI: A Web Server for Predicting Molecular Targets and Pathways of Cannabis-Based Therapeutics.Res Sq [Preprint]. 2024 Aug 9:rs.3.rs-4744915. doi: 10.21203/rs.3.rs-4744915/v1. Res Sq. 2024. Update in: J Cannabis Res. 2025 Feb 27;7(1):13. doi: 10.1186/s42238-025-00268-w. PMID: 39149470 Free PMC article. Updated. Preprint.
-
High Cannabigerol Hemp Extract Moderates Colitis and Modulates the Microbiome in an Inflammatory Bowel Disease Model.J Pharmacol Exp Ther. 2024 Aug 19;390(3):331-341. doi: 10.1124/jpet.124.002204. J Pharmacol Exp Ther. 2024. PMID: 39009468 Free PMC article.
-
The Use of Compounds Derived from Cannabis sativa in the Treatment of Epilepsy, Painful Conditions, and Neuropsychiatric and Neurodegenerative Disorders.Int J Mol Sci. 2024 May 25;25(11):5749. doi: 10.3390/ijms25115749. Int J Mol Sci. 2024. PMID: 38891938 Free PMC article. Review.
-
In-vitro Antioxidant and In-vivo Analgesics and Anti-inflammatory Activity of Allamanda blanchetii Leaf Extract in Rats.Antiinflamm Antiallergy Agents Med Chem. 2025;24(2):114-126. doi: 10.2174/0118715230340178241008163943. Antiinflamm Antiallergy Agents Med Chem. 2025. PMID: 39681830
References
-
- Boudreau D., Von Korff M., Rutter C.M., Saunders K., Ray G.T., Sullivan M.D., Campbell C.I., Merrill J.O., Silverberg M.J., Banta-Green C., et al. Trends in long-term opioid therapy for chronic non-cancer pain. Pharmacoepidemiol. Drug Saf. 2009;18:1166–1175. doi: 10.1002/pds.1833. - DOI - PMC - PubMed
-
- Campbell C.I., Weisner C., Leresche L., Ray G.T., Saunders K., Sullivan M.D., Banta-Green C.J., Merrill J.O., Silverberg M.J., Boudreau D., et al. Age and gender trends in long-term opioid analgesic use for noncancer pain. Am. J. Public Health. 2010;100:2541–2547. doi: 10.2105/AJPH.2009.180646. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

