Comparative Transcriptomic Analysis of Cerebellar Astrocytes across Developmental Stages and Brain Regions
- PMID: 38256095
- PMCID: PMC10816327
- DOI: 10.3390/ijms25021021
Comparative Transcriptomic Analysis of Cerebellar Astrocytes across Developmental Stages and Brain Regions
Abstract
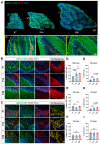
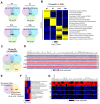
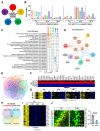
Astrocytes are the most abundant glial cell type in the central nervous system, and they play a crucial role in normal brain function. While gliogenesis and glial differentiation occur during perinatal cerebellar development, the processes that occur during early postnatal development remain obscure. In this study, we conducted transcriptomic profiling of postnatal cerebellar astrocytes at postnatal days 1, 7, 14, and 28 (P1, P7, P14, and P28), identifying temporal-specific gene signatures at each specific time point. Comparing these profiles with region-specific astrocyte differentially expressed genes (DEGs) published for the cortex, hippocampus, and olfactory bulb revealed cerebellar-specific gene signature across these developmental timepoints. Moreover, we conducted a comparative analysis of cerebellar astrocyte gene signatures with gene lists from pediatric brain tumors of cerebellar origin, including ependymoma and medulloblastoma. Notably, genes downregulated at P14, such as Kif11 and HMGB2, exhibited significant enrichment across all pediatric brain tumor groups, suggesting the importance of astrocytic gene repression during cerebellar development to these tumor subtypes. Collectively, our studies describe gene expression patterns during cerebellar astrocyte development, with potential implications for pediatric tumors originating in the cerebellum.
Keywords: astrocyte; cerebellum; pediatric brain tumors; postnatal development; transcriptome.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

