A Novel Combined Dry Powder Inhaler Comprising Nanosized Ketoprofen-Embedded Mannitol-Coated Microparticles for Pulmonary Inflammations: Development, In Vitro-In Silico Characterization, and Cell Line Evaluation
- PMID: 38256908
- PMCID: PMC10818896
- DOI: 10.3390/ph17010075
A Novel Combined Dry Powder Inhaler Comprising Nanosized Ketoprofen-Embedded Mannitol-Coated Microparticles for Pulmonary Inflammations: Development, In Vitro-In Silico Characterization, and Cell Line Evaluation
Abstract
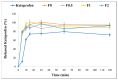
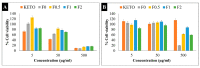
Pulmonary inflammations such as chronic obstructive pulmonary disease and cystic fibrosis are widespread and can be fatal, especially when they are characterized by abnormal mucus accumulation. Inhaled corticosteroids are commonly used for lung inflammations despite their considerable side effects. By utilizing particle engineering techniques, a combined dry powder inhaler (DPI) comprising nanosized ketoprofen-embedded mannitol-coated microparticles was developed. A nanoembedded microparticle system means a novel advance in pulmonary delivery by enhancing local pulmonary deposition while avoiding clearance mechanisms. Ketoprofen, a poorly water-soluble anti-inflammatory drug, was dispersed in the stabilizer solution and then homogenized by ultraturrax. Following this, a ketoprofen-containing nanosuspension was produced by wet-media milling. Furthermore, co-spray drying was conducted with L-leucine (dispersity enhancer) and mannitol (coating and mucuactive agent). Particle size, morphology, dissolution, permeation, viscosity, in vitro and in silico deposition, cytotoxicity, and anti-inflammatory effect were investigated. The particle size of the ketoprofen-containing nanosuspension was ~230 nm. SEM images of the spray-dried powder displayed wrinkled, coated, and nearly spherical particles with a final size of ~2 µm (nano-in-micro), which is optimal for pulmonary delivery. The mannitol-containing samples decreased the viscosity of 10% mucin solution. The results of the mass median aerodynamic diameter (2.4-4.5 µm), fine particle fraction (56-71%), permeation (five-fold enhancement), and dissolution (80% release in 5 min) confirmed that the system is ideal for local inhalation. All samples showed a significant anti-inflammatory effect and decreased IL-6 on the LPS-treated U937 cell line with low cytotoxicity. Hence, developing an innovative combined DPI comprising ketoprofen and mannitol by employing a nano-in-micro approach is a potential treatment for lung inflammations.
Keywords: combination product; inflammation; ketoprofen; mannitol; milling; nano-in-micro; particle engineering; pulmonary delivery; spray-drying.
Conflict of interest statement
The authors declare no conflicts of interest. The authors alone are responsible for the content and writing of this article.
Figures





Similar articles
-
Development of extra-fine particles containing nanosized meloxicam for deep pulmonary delivery: In vitro aerodynamic and cell line measurements.Eur J Pharm Sci. 2022 Sep 1;176:106247. doi: 10.1016/j.ejps.2022.106247. Epub 2022 Jun 25. Eur J Pharm Sci. 2022. PMID: 35760279
-
Preparation and Characterization of Ibuprofen Containing Nano-Embedded-Microparticles for Pulmonary Delivery.Pharmaceutics. 2023 Feb 6;15(2):545. doi: 10.3390/pharmaceutics15020545. Pharmaceutics. 2023. PMID: 36839867 Free PMC article.
-
Co-Spray Dried Mannitol/Poly(amidoamine)-Doxorubicin Dry-Powder Inhaler Formulations for Lung Adenocarcinoma: Morphology, In Vitro Evaluation, and Aerodynamic Performance.AAPS PharmSciTech. 2018 Feb;19(2):531-540. doi: 10.1208/s12249-017-0859-1. Epub 2017 Aug 23. AAPS PharmSciTech. 2018. PMID: 28840529
-
Mannitol-leucine synergy in nanocrystal agglomerates for enhanced systemic delivery of inhaled ketoprofen: Pharmacokinetics and safety in ovalbumin-sensitized rats.Int J Pharm. 2025 May 15;676:125610. doi: 10.1016/j.ijpharm.2025.125610. Epub 2025 Apr 19. Int J Pharm. 2025. PMID: 40258502
-
Non-steroidal anti-inflammatory drug for pulmonary administration: design and investigation of ketoprofen lysinate fine dry powders.Int J Pharm. 2013 May 1;448(1):198-204. doi: 10.1016/j.ijpharm.2013.03.030. Epub 2013 Mar 23. Int J Pharm. 2013. PMID: 23528281
Cited by
-
Diclofenac-Loaded Orodispersible Nanofibers Prepared by Double-Needle Electrospinning.Polymers (Basel). 2025 May 6;17(9):1262. doi: 10.3390/polym17091262. Polymers (Basel). 2025. PMID: 40363045 Free PMC article.
-
Encapsulation of Nanocrystals in Mannitol-Based Inhalable Microparticles via Spray-Drying: A Promising Strategy for Lung Delivery of Curcumin.Pharmaceuticals (Basel). 2024 Dec 18;17(12):1708. doi: 10.3390/ph17121708. Pharmaceuticals (Basel). 2024. PMID: 39770549 Free PMC article.
-
Lung Delivery of Lactose-Free Microparticles Loaded with Azithromycin for the Treatment of Bacterial Infections.Pharmaceutics. 2025 Jun 11;17(6):770. doi: 10.3390/pharmaceutics17060770. Pharmaceutics. 2025. PMID: 40574082 Free PMC article.
-
Fabrication of Ciprofloxacin-Loaded Sodium Alginate Nanobeads Coated with Thiol-Anchored Chitosan Using B-390 Encapsulator Following Optimization by DoE.Pharmaceutics. 2024 May 21;16(6):691. doi: 10.3390/pharmaceutics16060691. Pharmaceutics. 2024. PMID: 38931815 Free PMC article.
-
Optimization, In Vitro, and In Silico Characterization of Theophylline Inhalable Powder Using Raffinose-Amino Acid Combination as Fine Co-Spray-Dried Carriers.Pharmaceutics. 2025 Apr 3;17(4):466. doi: 10.3390/pharmaceutics17040466. Pharmaceutics. 2025. PMID: 40284461 Free PMC article.
References
-
- Fabbri L.M., Romagnoli M., Corbetta L., Casoni G., Busljetic K., Turato G., Ligabue G., Ciaccia A., Saetta M., Papi A. differences in airway inflammation in patients with fixed airflow obstruction due to asthma or chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2003;167:418–424. doi: 10.1164/rccm.200203-183OC. - DOI - PubMed
-
- Cesta M.C., Zippoli M., Marsiglia C., Gavioli E.M., Mantelli F., Allegretti M., Balk R.A. The Role of Interleukin-8 in Lung Inflammation and Injury: Implications for the Management of COVID-19 and Hyperinflammatory Acute Respiratory Distress Syndrome. Front. Pharmacol. 2021;12:808797. doi: 10.3389/fphar.2021.808797. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

