Towards More Precise Targeting of Inhaled Aerosols to Different Areas of the Respiratory System
- PMID: 38258107
- PMCID: PMC10818612
- DOI: 10.3390/pharmaceutics16010097
Towards More Precise Targeting of Inhaled Aerosols to Different Areas of the Respiratory System
Abstract
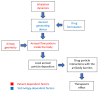
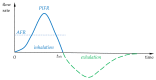
Pharmaceutical aerosols play a key role in the treatment of lung disorders, but also systemic diseases, due to their ability to target specific areas of the respiratory system (RS). This article focuses on identifying and clarifying the influence of various factors involved in the generation of aerosol micro- and nanoparticles on their regional distribution and deposition in the RS. Attention is given to the importance of process parameters during the aerosolization of liquids or powders and the role of aerosol flow dynamics in the RS. The interaction of deposited particles with the fluid environment of the lung is also pointed out as an important step in the mass transfer of the drug to the RS surface. The analysis presented highlights the technical aspects of preparing the precursors to ensure that the properties of the aerosol are suitable for a given therapeutic target. Through an analysis of existing technical limitations, selected strategies aimed at enhancing the effectiveness of targeted aerosol delivery to the RS have been identified and presented. These strategies also include the use of smart inhaling devices and systems with built-in AI algorithms.
Keywords: AI in drug delivery; aerosol; airflow dynamics; inhalation; nebulization; particle deposition; particle–lung interactions; smart inhalers.
Conflict of interest statement
The author declares no conflicts of interest.
Figures



References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

