Human B cells and dendritic cells are susceptible and permissive to enterovirus D68 infection
- PMID: 38259063
- PMCID: PMC10900886
- DOI: 10.1128/msphere.00526-23
Human B cells and dendritic cells are susceptible and permissive to enterovirus D68 infection
Abstract
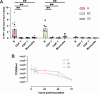
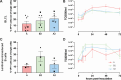
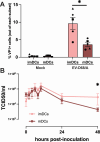
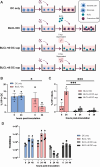
Enterovirus D68 (EV-D68) is predominantly associated with mild respiratory infections, but can also cause severe respiratory disease and extra-respiratory complications, including acute flaccid myelitis. Systemic dissemination of EV-D68 is crucial for the development of extra-respiratory diseases, but it is currently unclear how EV-D68 spreads systemically (viremia). We hypothesize that immune cells contribute to the systemic dissemination of EV-D68, as this is a mechanism commonly used by other enteroviruses. Therefore, we investigated the susceptibility and permissiveness of human primary immune cells for different EV-D68 isolates. In human peripheral blood mononuclear cells inoculated with EV-D68, only B cells were susceptible but virus replication was limited. However, in B cell-rich cultures, such as Epstein-Barr virus-transformed B-lymphoblastoid cell line (BLCL) and primary lentivirus-transduced B cells, which better represent lymphoid B cells, were productively infected. Subsequently, we showed that dendritic cells (DCs), particularly immature DCs, are susceptible and permissive for EV-D68 infection and that they can spread EV-D68 to autologous BLCL. Altogether, our findings suggest that immune cells, especially B cells and DCs, could play an important role in the pathogenesis of EV-D68 infection. Infection of these cells may contribute to systemic dissemination of EV-D68, which is an essential step toward the development of extra-respiratory complications.IMPORTANCEEnterovirus D68 (EV-D68) is an emerging respiratory virus that has caused outbreaks worldwide since 2014. EV-D68 infects primarily respiratory epithelial cells resulting in mild respiratory diseases. However, EV-D68 infection is also associated with extra-respiratory complications, including polio-like paralysis. It is unclear how EV-D68 spreads systemically and infects other organs. We hypothesized that immune cells could play a role in the extra-respiratory spread of EV-D68. We showed that EV-D68 can infect and replicate in specific immune cells, that is, B cells and dendritic cells (DCs), and that virus could be transferred from DCs to B cells. Our data reveal a potential role of immune cells in the pathogenesis of EV-D68 infection. Intervention strategies that prevent EV-D68 infection of immune cells will therefore potentially prevent systemic spread of virus and thereby severe extra-respiratory complications.
Keywords: B cells; dendritic cells; enterovirus; enterovirus D68; extra-respiratory infection; immune cells; systemic spread; viral pathogenesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
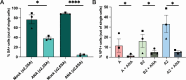


References
-
- Greninger AL, Naccache SN, Messacar K, Clayton A, Yu G, Somasekar S, Federman S, Stryke D, Anderson C, Yagi S, Messenger S, Wadford D, Xia D, Watt JP, Van Haren K, Dominguez SR, Glaser C, Aldrovandi G, Chiu CY. 2015. A novel outbreak enterovirus D68 strain associated with acute flaccid myelitis cases in the USA (2012-14): a retrospective cohort study. Lancet Infect Dis 15:671–682. doi: 10.1016/S1473-3099(15)70093-9 - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

