Impact of structural modifications of IgG antibodies on effector functions
- PMID: 38259472
- PMCID: PMC10800522
- DOI: 10.3389/fimmu.2023.1304365
Impact of structural modifications of IgG antibodies on effector functions
Abstract
Immunoglobulin G (IgG) antibodies are a critical component of the adaptive immune system, binding to and neutralizing pathogens and other foreign substances. Recent advances in molecular antibody biology and structural protein engineering enabled the modification of IgG antibodies to enhance their therapeutic potential. This review summarizes recent progress in both natural and engineered structural modifications of IgG antibodies, including allotypic variation, glycosylation, Fc engineering, and Fc gamma receptor binding optimization. We discuss the functional consequences of these modifications to highlight their potential for therapeutical applications.
Keywords: FcγR; IgG; allotypes; antibodies; complement; glycosylation; subclasses.
Copyright © 2024 Damelang, Brinkhaus, van Osch, Schuurman, Labrijn, Rispens and Vidarsson.
Conflict of interest statement
TD, JS, and AL were employed by and/or own warrants and/or stocks in Genmab. The work of MB was funded by argenx. JS and AL are inventors of patents/pending patent applications on technologies and mutations mentioned in this review. GV serves as a consultant for argenx. Genmab and argenx are both biotechnology companies that develop therapeutic antibodies. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


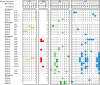
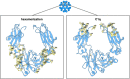

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

