Immunoseroproteomic profiling in autoantibody to ENO1 as potential biomarker in immunodiagnosis of osteosarcoma by serological proteome analysis (SERPA) approach
- PMID: 38260036
- PMCID: PMC10802918
- DOI: 10.1080/2162402X.2021.1966969
Immunoseroproteomic profiling in autoantibody to ENO1 as potential biomarker in immunodiagnosis of osteosarcoma by serological proteome analysis (SERPA) approach
Abstract
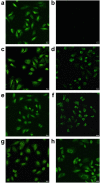
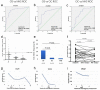
Osteosarcoma (OS) is the most common highly malignant primary solid bone tumor. Despite its relatively low incidence among cancers, it remains one of the most harmful primary malignant tumors in childhood and adolescence. It is now evident that serum autoantibodies against tumor-associated antigens (TAAs) could be used as serological cancer biomarkers in types of cancers. Serological proteome analysis (SERPA) approach was applied to profile anti-TAA autoantibody response in sera from patients with OS and normal human, as well as explore difference between this response. This approach can detect autoantibodies that could serve as clinical biomarkers and immunotherapeutic agents. Enzyme-linked immunosorbent assay (ELISA) and Western blotting were further used to validate the level of identified TAAs. ENO1 as a 47kD TAA in OS was identified and characterized by SERPA. Analysis of 172 serum samples with OS, osteochondroma (OC), and normal human sera (NHS) by ELISA showed higher frequency of anti-ENO1 autoantibodies in OS sera compared to others. Interestingly, decrease of ENO1 immunoreactivity was observed in most patients after treatments, which may imply a potential association between anti-ENO1 autoantibody titers and disease progression. Nine of twelve sera reacted strongly against purified ENO1, but three reacted weakly against purified ENO1, which indicated 75.0% sera with positive optimal density values from ELISA were consistently positive in Western blotting. The expression of ENO1 in OS tissues was evaluated by immunohistochemistry in tumor microarray. ENO1 was one of the autoantibodies that elicit autoimmune responses in OS and can be used as biomarkers in immunodiagnosis and progression of OS.
Keywords: ENO1; Osteosarcoma; immunodiagnosis; serological proteome analysis; tumor-associated antigen.
© 2021 The Author(s). Published with license by Taylor & Francis Group, LLC.
Conflict of interest statement
The authors report no conflict of interest.
Figures




Similar articles
-
Evaluating a Panel of Autoantibodies Against Tumor-Associated Antigens in Human Osteosarcoma.Front Genet. 2022 Apr 25;13:872253. doi: 10.3389/fgene.2022.872253. eCollection 2022. Front Genet. 2022. PMID: 35547257 Free PMC article.
-
Serological proteome analysis approach-based identification of ENO1 as a tumor-associated antigen and its autoantibody could enhance the sensitivity of CEA and CYFRA 21-1 in the detection of non-small cell lung cancer.Oncotarget. 2017 May 30;8(22):36664-36673. doi: 10.18632/oncotarget.17067. Oncotarget. 2017. PMID: 28456790 Free PMC article.
-
Immunoseroproteomic Profiling in African American Men with Prostate Cancer: Evidence for an Autoantibody Response to Glycolysis and Plasminogen-Associated Proteins.Mol Cell Proteomics. 2016 Dec;15(12):3564-3580. doi: 10.1074/mcp.M116.060244. Epub 2016 Oct 14. Mol Cell Proteomics. 2016. PMID: 27742740 Free PMC article.
-
Tumor-Associated Antigens (TAAs) for the Serological Diagnosis of Osteosarcoma.Front Immunol. 2021 Apr 30;12:665106. doi: 10.3389/fimmu.2021.665106. eCollection 2021. Front Immunol. 2021. PMID: 33995397 Free PMC article. Review.
-
Using immunoproteomics to identify tumor-associated antigens (TAAs) as biomarkers in cancer immunodiagnosis.Autoimmun Rev. 2013 Oct;12(12):1123-8. doi: 10.1016/j.autrev.2013.06.015. Epub 2013 Jun 24. Autoimmun Rev. 2013. PMID: 23806562 Free PMC article. Review.
Cited by
-
Evaluating a Panel of Autoantibodies Against Tumor-Associated Antigens in Human Osteosarcoma.Front Genet. 2022 Apr 25;13:872253. doi: 10.3389/fgene.2022.872253. eCollection 2022. Front Genet. 2022. PMID: 35547257 Free PMC article.
-
A 3D-printed scaffold-based osteosarcoma model allows to investigate tumor phenotypes and pathogenesis in an in vitro bone-mimicking niche.Mater Today Bio. 2022 May 25;15:100295. doi: 10.1016/j.mtbio.2022.100295. eCollection 2022 Jun. Mater Today Bio. 2022. PMID: 35665234 Free PMC article.
-
Role of ENO1 and its targeted therapy in tumors.J Transl Med. 2024 Nov 14;22(1):1025. doi: 10.1186/s12967-024-05847-8. J Transl Med. 2024. PMID: 39543641 Free PMC article. Review.
References
-
- Huvos AG. Bone tumors: diagnosis, treatment, and prognosis. Second ed. Philadelphia: Saunders; New York, USA, 1991. ISBN:0-7216-2050-7.
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
