This is a preprint.
TRPV4 is expressed by enteric glia and muscularis macrophages of the colon but does not play a prominent role in colonic motility
- PMID: 38260314
- PMCID: PMC10802399
- DOI: 10.1101/2024.01.09.574831
TRPV4 is expressed by enteric glia and muscularis macrophages of the colon but does not play a prominent role in colonic motility
Abstract
Background: Mechanosensation is an important trigger of physiological processes in the gastrointestinal tract. Aberrant responses to mechanical input are associated with digestive disorders, including visceral hypersensitivity. Transient Receptor Potential Vanilloid 4 (TRPV4) is a mechanosensory ion channel with proposed roles in visceral afferent signaling, intestinal inflammation, and gut motility. While TRPV4 is a potential therapeutic target for digestive disease, current mechanistic understanding of how TRPV4 may influence gut function is limited by inconsistent reports of TRPV4 expression and distribution.
Methods: In this study we profiled functional expression of TRPV4 using Ca2+ imaging of wholemount preparations of the mouse, monkey, and human intestine in combination with immunofluorescent labeling for established cellular markers. The involvement of TRPV4 in colonic motility was assessed in vitro using videomapping and contraction assays.
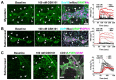
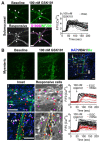
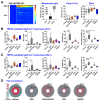
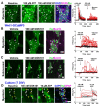
Results: The TRPV4 agonist GSK1016790A evoked Ca2+ signaling in muscularis macrophages, enteric glia, and endothelial cells. TRPV4 specificity was confirmed using TRPV4 KO mouse tissue or antagonist pre-treatment. Calcium responses were not detected in other cell types required for neuromuscular signaling including enteric neurons, interstitial cells of Cajal, PDGFRα+ cells, and intestinal smooth muscle. TRPV4 activation led to rapid Ca2+ responses by a subpopulation of glial cells, followed by sustained Ca2+ signaling throughout the enteric glial network. Propagation of these waves was suppressed by inhibition of gap junctions or Ca2+ release from intracellular stores. Coordinated glial signaling in response to GSK1016790A was also disrupted in acute TNBS colitis. The involvement of TRPV4 in the initiation and propagation of colonic motility patterns was examined in vitro.
Conclusions: We reveal a previously unappreciated role for TRPV4 in the initiation of distension-evoked colonic motility. These observations provide new insights into the functional role of TRPV4 activation in the gut, with important implications for how TRPV4 may influence critical processes including inflammatory signaling and motility.
Keywords: Bioimage analysis; Calcium Imaging; Enteric Glial Cells; Motility; Resident Macrophages; TRPV4; Transient Receptor Potential.
Figures


Similar articles
-
Transient receptor potential vanilloid 4 inhibits mouse colonic motility by activating NO-dependent enteric neurotransmission.J Mol Med (Berl). 2015 Dec;93(12):1297-309. doi: 10.1007/s00109-015-1336-5. Epub 2015 Sep 2. J Mol Med (Berl). 2015. PMID: 26330151
-
Agonist-evoked Ca2+ signaling in enteric glia drives neural programs that regulate intestinal motility in mice.Cell Mol Gastroenterol Hepatol. 2015 Nov 1;1(6):631-645. doi: 10.1016/j.jcmgh.2015.08.004. Cell Mol Gastroenterol Hepatol. 2015. PMID: 26693173 Free PMC article.
-
TRPV4 stimulates colonic afferents through mucosal release of ATP and glutamate.Br J Pharmacol. 2025 Mar;182(6):1324-1340. doi: 10.1111/bph.17408. Epub 2024 Dec 3. Br J Pharmacol. 2025. PMID: 39626870
-
The Role of the TRPV4 Channel in Intestinal Physiology and Pathology.J Inflamm Res. 2024 Nov 21;17:9307-9317. doi: 10.2147/JIR.S483350. eCollection 2024. J Inflamm Res. 2024. PMID: 39588136 Free PMC article. Review.
-
Enteric glia as a player of gut-brain interactions during Parkinson's disease.Front Neurosci. 2023 Nov 1;17:1281710. doi: 10.3389/fnins.2023.1281710. eCollection 2023. Front Neurosci. 2023. PMID: 38027511 Free PMC article. Review.
References
-
- Spencer NJ, Zagorodnyuk V, Brookes SJ, Hibberd T. Spinal afferent nerve endings in visceral organs: recent advances. Am J Physiol Gastrointest Liver Physiol 2016;311:G1056–G63. - PubMed
-
- Cenac N, Altier C, Chapman K, Liedtke W, Zamponi G, Vergnolle N. Transient receptor potential vanilloid-4 has a major role in visceral hypersensitivity symptoms. Gastroenterology 2008;135:937–46, 46 e1-2. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
