A Minimally Invasive Robotic Tissue Palpation Device
- PMID: 38261510
- PMCID: PMC11178256
- DOI: 10.1109/TBME.2024.3357293
A Minimally Invasive Robotic Tissue Palpation Device
Abstract
Objective: Robot-assisted minimally invasive surgery remains limited by the absence of haptic feedback, which surgeons routinely rely on to assess tissue stiffness. This limitation hinders surgeons' ability to identify and treat abnormal tissues, such as tumors, during robotic surgery.
Methods: To address this challenge, we developed a robotic tissue palpation device capable of rapidly and non-invasively quantifying the stiffness of soft tissues, allowing surgeons to make objective and data-driven decisions during minimally invasive procedures. We evaluated the effectiveness of our device by measuring the stiffness of phantoms as well as lung, heart, liver, and skin tissues obtained from both rats and swine.
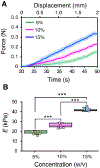
Results: Results demonstrated that our device can accurately determine tissue stiffness and identify tumor mimics. Specifically, in swine lung, we determined elastic modulus (E) values of 9.1 ± 2.3, 16.8 ± 1.8, and 26.0 ± 3.6 kPa under different internal pressure of the lungs (PIP) of 2, 25, and 45 cmH2O, respectively. Using our device, we successfully located a 2-cm tumor mimic embedded at a depth of 5 mm in the lung subpleural region. Additionally, we measured E values of 33.0 ± 5.4, 19.2 ± 2.2, 33.5 ± 8.2, and 22.6 ± 6.0 kPa for swine heart, liver, abdominal skin, and muscle, respectively, which closely matched existing literature data.
Conclusion/significance: Results suggest that our robotic palpation device can be utilized during surgery, either as a stand-alone or additional tool integrated into existing robotic surgical systems, to enhance treatment outcomes by enabling accurate intraoperative identification of abnormal tissue.
Figures

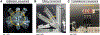

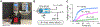


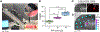

Similar articles
-
Artificial palpation in robotic surgery using haptic feedback.Surg Endosc. 2019 Apr;33(4):1252-1259. doi: 10.1007/s00464-018-6405-8. Epub 2018 Sep 5. Surg Endosc. 2019. PMID: 30187198 Free PMC article.
-
Using visual cues to enhance haptic feedback for palpation on virtual model of soft tissue.Med Biol Eng Comput. 2015 Nov;53(11):1177-86. doi: 10.1007/s11517-015-1309-4. Epub 2015 May 28. Med Biol Eng Comput. 2015. PMID: 26018755
-
Effects of realistic force feedback in a robotic assisted minimally invasive surgery system.Minim Invasive Ther Allied Technol. 2014 Jun;23(3):127-35. doi: 10.3109/13645706.2013.867886. Epub 2013 Dec 12. Minim Invasive Ther Allied Technol. 2014. PMID: 24328984
-
Robotic natural orifice transluminal endoscopic surgery (R-NOTES): literature review and prototype system.Minim Invasive Ther Allied Technol. 2015 Feb;24(1):18-23. doi: 10.3109/13645706.2014.992907. Epub 2014 Dec 25. Minim Invasive Ther Allied Technol. 2015. PMID: 25539996 Review.
-
Technical approach of robotic total right hepatic lobectomy: How we do it?J Robot Surg. 2019 Apr;13(2):193-199. doi: 10.1007/s11701-018-0881-7. Epub 2018 Oct 1. J Robot Surg. 2019. PMID: 30276634 Review.
Cited by
-
A Tension Sensor Array for Cable-Driven Surgical Robots.Sensors (Basel). 2024 May 16;24(10):3156. doi: 10.3390/s24103156. Sensors (Basel). 2024. PMID: 38794010 Free PMC article.
-
Bioimpedance measurements of fibrotic and acutely injured lung tissues.Acta Biomater. 2025 Mar 1;194:270-287. doi: 10.1016/j.actbio.2025.01.039. Epub 2025 Jan 25. Acta Biomater. 2025. PMID: 39870150
References
-
- Peters BS et al., “Review of emerging surgical robotic technology,” Surg Endosc, vol. 32, no. 4, pp. 1636–1655, Apr, 2018. - PubMed
-
- Vitiello V et al., “Emerging Robotic Platforms for Minimally Invasive Surgery,” IEEE Reviews in Biomedical Engineering, vol. 6, pp. 111–126, 2013. - PubMed
-
- Omisore OM et al., “A Review on Flexible Robotic Systems for Minimally Invasive Surgery,” IEEE Transactions on Systems, Man, and Cybernetics: Systems, vol. 52, no. 1, pp. 631–644, 2022.
-
- Kim U et al., “A Surgical Palpation Probe With 6-Axis Force/Torque Sensing Capability for Minimally Invasive Surgery,” IEEE Transactions on Industrial Electronics, vol. 65, pp. 2755–2765, 2018.
-
- Ibrahim MK et al., “Robot-assisted for medical surgery: A literature review,” AIP Conference Proceedings, vol. 2591, no. 1, pp. 030008, 2023/03/29, 2023.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

