The perspective of cAMP/cGMP signaling and cyclic nucleotide phosphodiesterases in aortic aneurysm and dissection
- PMID: 38262506
- PMCID: PMC10939884
- DOI: 10.1016/j.vph.2024.107278
The perspective of cAMP/cGMP signaling and cyclic nucleotide phosphodiesterases in aortic aneurysm and dissection
Abstract
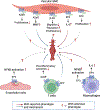
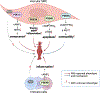
Aortic aneurysm (AA) and dissection (AD) are aortic diseases caused primarily by medial layer degeneration and perivascular inflammation. They are lethal when the rupture happens. Vascular smooth muscle cells (SMCs) play critical roles in the pathogenesis of medial degeneration, characterized by SMC loss and elastin fiber degradation. Many molecular pathways, including cyclic nucleotide signaling, have been reported in regulating vascular SMC functions, matrix remodeling, and vascular structure integrity. Intracellular cyclic adenosine monophosphate (cAMP) and cyclic guanosine monophosphate (cGMP) are second messengers that mediate intracellular signaling transduction through activating effectors, such as protein kinase A (PKA) and PKG, respectively. cAMP and cGMP are synthesized by adenylyl cyclase (AC) and guanylyl cyclase (GC), respectively, and degraded by cyclic nucleotide phosphodiesterases (PDEs). In this review, we will discuss the roles and mechanisms of cAMP/cGMP signaling and PDEs in AA/AD formation and progression and the potential of PDE inhibitors in AA/AD, whether they are beneficial or detrimental. We also performed database analysis and summarized the results showing PDEs with significant expression changes under AA/AD, which should provide rationales for future research on PDEs in AA/AD.
Keywords: Aortic aneurysm; Aortic dissection; GPCR; PDE; cAMP; cGMP.
Copyright © 2024 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declared no conflict of interests.
Figures

Similar articles
-
Roles of phosphodiesterases in the regulation of the cardiac cyclic nucleotide cross-talk signaling network.J Mol Cell Cardiol. 2016 Feb;91:215-27. doi: 10.1016/j.yjmcc.2016.01.004. Epub 2016 Jan 7. J Mol Cell Cardiol. 2016. PMID: 26773602 Free PMC article.
-
Diverse Roles of the Multiple Phosphodiesterases in the Regulation of Cyclic Nucleotide Signaling in Dictyostelium.Cells. 2025 Apr 1;14(7):522. doi: 10.3390/cells14070522. Cells. 2025. PMID: 40214475 Free PMC article. Review.
-
Cyclic nucleotide signaling changes associated with normal aging and age-related diseases of the brain.Cell Signal. 2018 Jan;42:281-291. doi: 10.1016/j.cellsig.2017.11.004. Epub 2017 Nov 23. Cell Signal. 2018. PMID: 29175000 Free PMC article. Review.
-
Cyclic nucleotide signalling compartmentation by PDEs in cultured vascular smooth muscle cells.Br J Pharmacol. 2019 Jun;176(11):1780-1792. doi: 10.1111/bph.14651. Epub 2019 Apr 24. Br J Pharmacol. 2019. PMID: 30825186 Free PMC article.
-
Phosphodiesterase in heart and vessels: from physiology to diseases.Physiol Rev. 2024 Apr 1;104(2):765-834. doi: 10.1152/physrev.00015.2023. Epub 2023 Nov 16. Physiol Rev. 2024. PMID: 37971403 Free PMC article. Review.
Cited by
-
Identification of Key Nucleotide Metabolism Genes in Diabetic Retinopathy Based on Bioinformatics Analysis and Experimental Verification.Biology (Basel). 2025 Apr 12;14(4):409. doi: 10.3390/biology14040409. Biology (Basel). 2025. PMID: 40282274 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

