High-dose amphotericin: yay or nay? A case series and literature review
- PMID: 38264401
- PMCID: PMC10803125
- DOI: 10.7573/dic.2023-9-1
High-dose amphotericin: yay or nay? A case series and literature review
Abstract
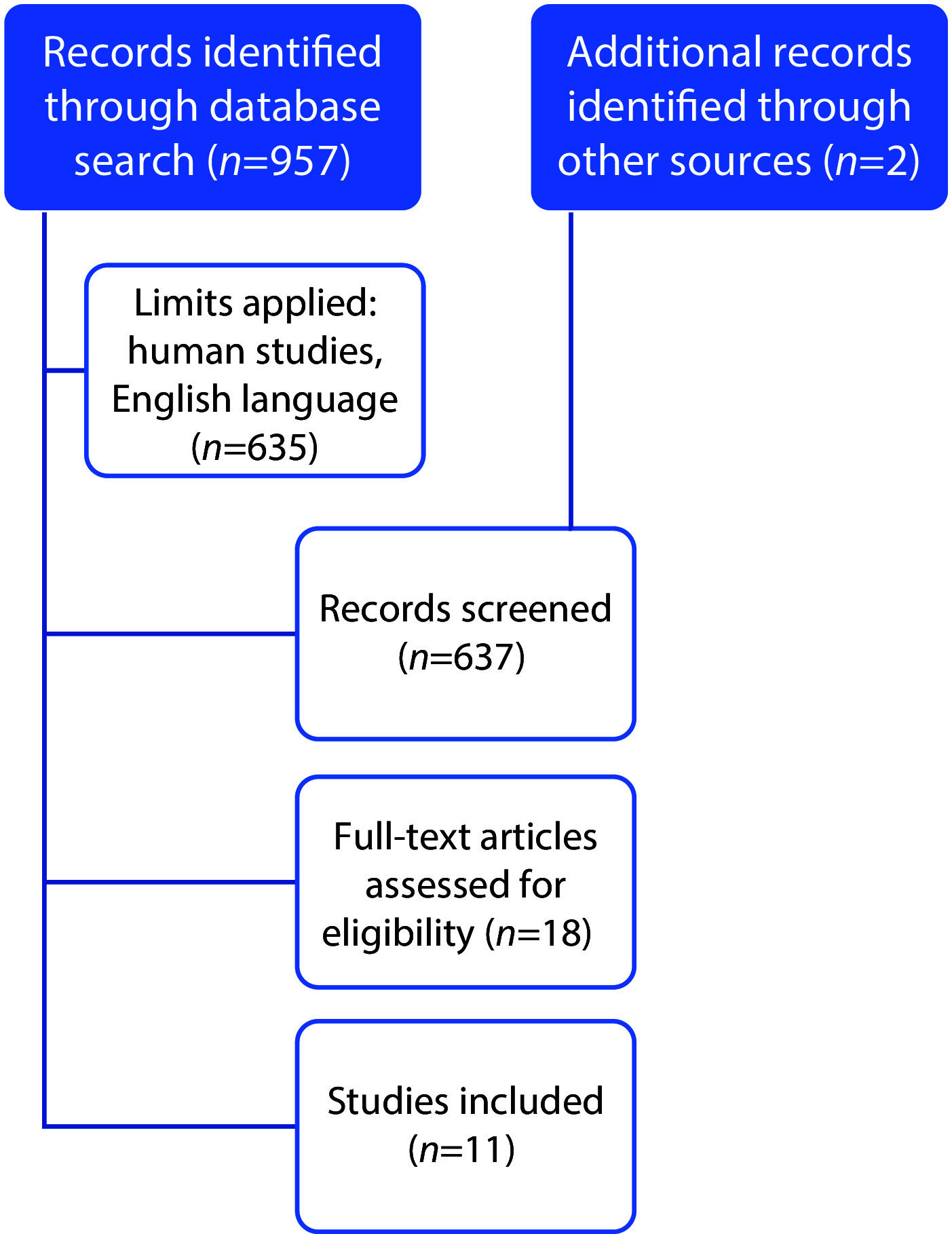
Invasive fungal infections pose significant morbidity and mortality risks, particularly those caused by moulds. Available antifungal classes are limited by toxicities and are increasingly susceptible to resistance, particularly amongst challenging fungal pathogens. The purpose of this case series and literature review was to characterize the use of a high-dose lipid formulation of amphotericin B. A case series is presented including patients who received high-dose lipid formulation amphotericin B (≥7.5 mg/kg/day) between June 2012 and August 2021. Additionally, a systematic literature review was conducted by searching the PubMed database for English-language studies involving individuals who received high-dose amphotericin B therapy (≥7.5 mg/kg) using lipid formulations. Nine patients were included in the case series, receiving an average of 8.9 ± 1.3 mg/kg liposomal amphotericin B over a mean of 11.0 ± 10.8 days predominantly for mould infections including Mucorales, aspergillosis and Fusarium. The patients were primarily cared for in intensive care units, with varying treatment histories and outcomes. A total of 11 studies (n=260 patients) met inclusion criteria for the literature review. Responses to high-dose liposomal amphotericin B ranged from 8% to 100%, often showing favourable outcomes. High doses of liposomal amphotericin B were well tolerated both in the case series and in published literature, with serum creatinine changes being the most commonly reported adverse event. However, multi-patient studies continue to report less than favourable (range 8-62%) response rates. High-dose liposomal amphotericin B, either alone or in combination with other antifungal agents, might be a viable strategy for managing invasive fungal infections when few treatment choices exist. This article is part of the Challenges and strategies in the management of invasive fungal infections Special Issue: https://www.drugsincontext.com/special_issues/challenges-and-strategies-in-the-management-of-invasive-fungal-infections.
Keywords: amphotericin; antifungal agents; drug resistance; fungal; maximally tolerated dose.
Copyright © 2024 Stover KR, Jordan TE, Wagner JL, Barber KE.
Conflict of interest statement
Disclosure and potential conflicts of interest: KS has served as an advisory panel member for Cidara Therapeutics, Inc. and is currently serving as the American College of Clinical Pharmacy Secretary. All other authors have nothing to disclose. The International Committee of Medical Journal Editors (ICMJE) Potential Conflicts of Interests form for the authors is available for download at: https://www.drugsincontext.com/wp-content/uploads/2023/12/dic.2023-9-1-COI.pdf
References
-
- Donnelly JP, Chen SC, Kauffman CA, et al. Revision and update of the consensus definitions of invasive fungal disease from the European Organization for Research and Treatment of Cancer and the mycoses study group education and research consortium. Clin Infect Dis. 2020;71(6):1367–1376. doi: 10.1093/cid/ciz1008. - DOI - PMC - PubMed
-
- Bupha-Intr O, Butters C, Reynolds G, et al. Australasian Antifungal Guidelines Steering Committee. Consensus guidelines for the diagnosis and management of invasive fungal disease due to moulds other than Aspergillus in the haematology/oncology setting, 2021. Intern Med J. 2021;51(Suppl 7):177–219. doi: 10.1111/imj.15592. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous