EZH2 Inhibition Promotes Tumor Immunogenicity in Lung Squamous Cell Carcinomas
- PMID: 38265267
- PMCID: PMC10863487
- DOI: 10.1158/2767-9764.CRC-23-0399
EZH2 Inhibition Promotes Tumor Immunogenicity in Lung Squamous Cell Carcinomas
Abstract
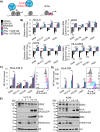
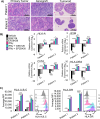
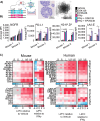
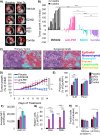
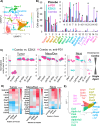
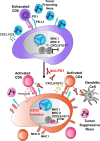
Two important factors that contribute to resistance to immune checkpoint inhibitors (ICI) are an immune-suppressive microenvironment and limited antigen presentation by tumor cells. In this study, we examine whether inhibition of the methyltransferase enhancer of zeste 2 (EZH2) can increase ICI response in lung squamous cell carcinomas (LSCC). Our in vitro experiments using two-dimensional human cancer cell lines as well as three-dimensional murine and patient-derived organoids treated with two inhibitors of the EZH2 plus IFNγ showed that EZH2 inhibition leads to expression of both MHC class I and II (MHCI/II) expression at both the mRNA and protein levels. Chromatin immunoprecipitation sequencing confirmed loss of EZH2-mediated histone marks and gain of activating histone marks at key loci. Furthermore, we demonstrate strong tumor control in models of both autochthonous and syngeneic LSCC treated with anti-PD1 immunotherapy with EZH2 inhibition. Single-cell RNA sequencing and immune cell profiling demonstrated phenotypic changes toward more tumor suppressive phenotypes in EZH2 inhibitor-treated tumors. These results indicate that EZH2 inhibitors could increase ICI responses in patients undergoing treatment for LSCC.
Significance: The data described here show that inhibition of the epigenetic enzyme EZH2 allows derepression of multiple immunogenicity factors in LSCC, and that EZH2 inhibition alters myeloid cells in vivo. These data support clinical translation of this combination therapy for treatment of this deadly tumor type.
© 2024 The Authors; Published by the American Association for Cancer Research.
Figures

Update of
-
EZH2 inhibition promotes tumor immunogenicity in lung squamous cell carcinomas.bioRxiv [Preprint]. 2023 Jun 8:2023.06.06.543919. doi: 10.1101/2023.06.06.543919. bioRxiv. 2023. Update in: Cancer Res Commun. 2024 Feb 13;4(2):388-403. doi: 10.1158/2767-9764.CRC-23-0399. PMID: 37333199 Free PMC article. Updated. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

