A chimeric antigen receptor-based cellular safeguard mechanism for selective in vivo depletion of engineered T cells
- PMID: 38274808
- PMCID: PMC10808742
- DOI: 10.3389/fimmu.2023.1268698
A chimeric antigen receptor-based cellular safeguard mechanism for selective in vivo depletion of engineered T cells
Abstract
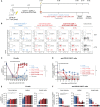
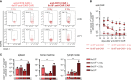
Adoptive immunotherapy based on chimeric antigen receptor (CAR)-engineered T cells has exhibited impressive clinical efficacy in treating B-cell malignancies. However, the potency of CAR-T cells carriethe potential for significant on-target/off-tumor toxicities when target antigens are shared with healthy cells, necessitating the development of complementary safety measures. In this context, there is a need to selectively eliminate therapeutically administered CAR-T cells, especially to revert long-term CAR-T cell-related side effects. To address this, we have developed an effective cellular-based safety mechanism to specifically target and eliminate the transferred CAR-T cells. As proof-of-principle, we have designed a secondary CAR (anti-CAR CAR) capable of recognizing a short peptide sequence (Strep-tag II) incorporated into the hinge domain of an anti-CD19 CAR. In in vitro experiments, these anti-CAR CAR-T cells have demonstrated antigen-specific cytokine release and cytotoxicity when co-cultured with anti-CD19 CAR-T cells. Moreover, in both immunocompromised and immunocompetent mice, we observed the successful depletion of anti-CD19 CAR-T cells when administered concurrently with anti-CAR CAR-T cells. We have also demonstrated the efficacy of this safeguard mechanism in a clinically relevant animal model of B-cell aplasia induced by CD19 CAR treatment, where this side effect was reversed upon anti-CAR CAR-T cells infusion. Notably, efficient B-cell recovery occurred even in the absence of any pre-conditioning regimens prior anti-CAR CAR-T cells transfer, thus enhancing its practical applicability. In summary, we developed a robust cellular safeguard system for selective in vivo elimination of engineered T cells, offering a promising solution to address CAR-T cell-related on-target/off-tumor toxicities.
Keywords: B cell aplasia 2; chimeric antigen receptor; on-target/off-tumor; safeguard mechanism; side effects.
Copyright © 2024 Svec, Dötsch, Warmuth, Trebo, Fräßle, Riddell, Jäger, D’Ippolito and Busch.
Conflict of interest statement
DB is co-founder of STAGE Cell Therapeutics GmbH now Juno Therapeutics, a Bristol-Myers Squibb Company and T Cell Factory B.V. now Kite/a Gilead Company. DB has a consulting contract with and receives sponsored research support from Juno Therapeutics/BMS. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- Sadelain M, Brentjens R, Rivière I. The basic principles of chimeric antigen receptor design. Cancer Discovery (2013) 3(4):388–98. doi: 10.1158/2159-8290.CD-12-0548 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

