Dynamic Imaging of Blood Coagulation Within the Hematoma of Patients With Acute Hemorrhagic Stroke
- PMID: 38275117
- PMCID: PMC10962442
- DOI: 10.1161/STROKEAHA.123.044343
Dynamic Imaging of Blood Coagulation Within the Hematoma of Patients With Acute Hemorrhagic Stroke
Abstract
Background: The dynamics of blood clot (combination of Hb [hemoglobin], fibrin, and a higher concentration of aggregated red blood cells) formation within the hematoma of an intracerebral hemorrhage is not well understood. A quantitative neuroimaging method of localized coagulated blood volume/distribution within the hematoma might improve clinical decision-making.
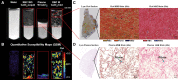
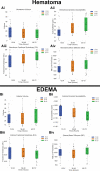
Methods: The deoxyhemoglobin of aggregated red blood cells within extravasated blood exhibits a higher magnetic susceptibility due to unpaired heme iron electrons. We propose that coagulated blood, with higher aggregated red blood cell content, will exhibit (1) a higher positive susceptibility than noncoagulated blood and (2) increase in fibrin polymerization-restricted localized diffusion, which can be measured noninvasively using quantitative susceptibility mapping and diffusion tensor imaging. In this serial magnetic resonance imaging study, we enrolled 24 patients with acute intracerebral hemorrhage between October 2021 to May 2022 at a stroke center. Patients were 30 to 70 years of age and had a hematoma volume >15 cm3 and National Institutes of Health Stroke Scale score >1. The patients underwent imaging 3×: within 12 to 24 (T1), 36 to 48 (T2), and 60 to 72 (T3) hours of last seen well on a 3T magnetic resonance imaging system. Three-dimensional anatomic, multigradient echo and 2-dimensional diffusion tensor images were obtained. Hematoma and edema volumes were calculated, and the distribution of coagulation was measured by dynamic changes in the susceptibilities and fractional anisotropy within the hematoma.
Results: Using a coagulated blood phantom, we demonstrated a linear relationship between the percentage coagulation and susceptibility (R2=0.91) with a positive red blood cell stain of the clot. The quantitative susceptibility maps showed a significant increase in hematoma susceptibility (T1, 0.29±0.04 parts per millions; T2, 0.36±0.04 parts per millions; T3, 0.45±0.04 parts per millions; P<0.0001). A concomitant increase in fractional anisotropy was also observed with time (T1, 0.40±0.02; T2, 0.45±0.02; T3, 0.47±0.02; P<0.05).
Conclusions: This quantitative neuroimaging study of coagulation within the hematoma has the potential to improve patient management, such as safe resumption of anticoagulants, the need for reversal agents, the administration of alteplase to resolve the clot, and the need for surgery.
Keywords: blood coagulation; cerebral hemorrhage; fibrin; magnetic resonance imaging; quantitative phase imaging.
Conflict of interest statement
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

