Identification of Virulence Factors in Isolates of Candida haemulonii, Candida albicans and Clavispora lusitaniae with Low Susceptibility and Resistance to Fluconazole and Amphotericin B
- PMID: 38276197
- PMCID: PMC10819056
- DOI: 10.3390/microorganisms12010212
Identification of Virulence Factors in Isolates of Candida haemulonii, Candida albicans and Clavispora lusitaniae with Low Susceptibility and Resistance to Fluconazole and Amphotericin B
Abstract
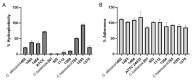
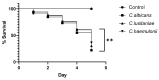
Emerging life-threatening multidrug-resistant (MDR) species such as the C. haemulonii species complex, Clavispora lusitaniae (sin. C. lusitaniae), and other Candida species are considered as an increasing risk for human health in the near future. (1) Background: Many studies have emphasized that the increase in drug resistance can be associated with several virulence factors in Candida and its knowledge is also essential in developing new antifungal strategies. (2) Methods: Hydrophobicity, adherence, biofilm formation, lipase activity, resistance to osmotic stress, and virulence 'in vivo' on G. mellonella larvae were studied in isolates of C. haemulonii, C. albicans, and C. lusitaniae with low susceptibility and resistance to fluconazole and amphotericin B. (3) Results: Intra- and interspecies variability were observed. C. haemulonii showed high hydrophobicity and the ability to adhere to and form biofilm. C. lusitaniae was less hydrophobic, was biofilm-formation-strain-dependent, and did not show lipase activity. Larvae inoculated with C. albicans isolates displayed significantly higher mortality rates than those infected with C. haemulonii and C. lusitaniae. (4) Conclusions: The ability to adhere to and form biofilms associated with their hydrophobic capacity, to adapt to stress, and to infect within an in vivo model, observed in these non-wild-type Candida and Clavispora isolates, shows their marked virulence features. Since factors that define virulence are related to the development of the resistance of these fungi to the few antifungals available for clinical use, differences in the physiology of these cells must be considered to develop new antifungal therapies.
Keywords: C. albicans; C. haemulonii; C. lusitaniae; Galleria mellonella; adherence; biofilm; hydrophobicity; resistance; stress; virulence factors.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
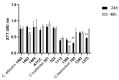
Similar articles
-
Virulence of Candida haemulonii complex in Galleria mellonella and efficacy of classical antifungal drugs: a comparative study with other clinically relevant non-albicans Candida species.FEMS Yeast Res. 2018 Nov 1;18(7). doi: 10.1093/femsyr/foy082. FEMS Yeast Res. 2018. PMID: 30052907
-
The Threat Called Candida haemulonii Species Complex in Rio de Janeiro State, Brazil: Focus on Antifungal Resistance and Virulence Attributes.J Fungi (Basel). 2022 May 27;8(6):574. doi: 10.3390/jof8060574. J Fungi (Basel). 2022. PMID: 35736057 Free PMC article.
-
Candida and candidaemia. Susceptibility and epidemiology.Dan Med J. 2013 Nov;60(11):B4698. Dan Med J. 2013. PMID: 24192246 Review.
-
Phenotypic and genotypic characterization of virulence markers and antifungal susceptibility of oral Candida species from diabetic and non-diabetic hemodialysis patients.BMC Oral Health. 2023 May 4;23(1):261. doi: 10.1186/s12903-023-02970-8. BMC Oral Health. 2023. PMID: 37143002 Free PMC article.
-
Non-albicans Candida spp. causing fungaemia: pathogenicity and antifungal resistance.J Hosp Infect. 2002 Apr;50(4):243-60. doi: 10.1053/jhin.2001.1151. J Hosp Infect. 2002. PMID: 12014897 Review.
Cited by
-
Fungal Empyema Thoracis Associated with Clavispora lusitaniae-First Report in a Domestic Cat.J Fungi (Basel). 2025 Feb 20;11(3):170. doi: 10.3390/jof11030170. J Fungi (Basel). 2025. PMID: 40137208 Free PMC article.
-
Virulence profile of pathogenic yeasts from snakes: Alternative ways for antifungal strategies.PLoS One. 2025 Mar 12;20(3):e0318703. doi: 10.1371/journal.pone.0318703. eCollection 2025. PLoS One. 2025. PMID: 40072936 Free PMC article.
-
Occurrence of pathogenic yeast species in artisanal cheeses from Boyacá, Colombia, including fluconazole resistant isolates.F1000Res. 2024 Oct 4;13:789. doi: 10.12688/f1000research.152447.3. eCollection 2024. F1000Res. 2024. PMID: 39464777 Free PMC article.
-
Deciphering cargo contents in extracellular vesicles of Candida haemulonii var. vulnera.Comput Struct Biotechnol J. 2025 Apr 15;27:1887-1900. doi: 10.1016/j.csbj.2025.04.014. eCollection 2025. Comput Struct Biotechnol J. 2025. PMID: 40487199 Free PMC article.
-
Characterization of Candida species isolated from clinical specimens: insights into virulence traits, antifungal resistance and molecular profiles.BMC Microbiol. 2024 Oct 5;24(1):388. doi: 10.1186/s12866-024-03515-x. BMC Microbiol. 2024. PMID: 39367309 Free PMC article.
References
-
- Hossain C.M., Ryan L.K., Gera M., Choudhuri S., Lyle N., Ali K.A., Diamond G. Antifungal and drug resistance. Encyclopedia. 2022;2:1722–1737. doi: 10.3390/encyclopedia2040118. - DOI
-
- Ramos L.S., Figueiredo-Carvalho M.H.G., Silva L.N., Siqueira N.L., Lima J.C., Oliveira S.S., Almeida-Paes R., Zancopé-Oliveira R.M., Azevedo F.S., Ferreira A.L., et al. The Threat Called Candida haemulonii Species Complex in Rio de Janeiro State, Brazil: Focus on Antifungal Resistance and Virulence Attributes. J. Fungi. 2022;8:574. doi: 10.3390/jof8060574. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

