Isopropanol production via the thermophilic bioconversion of sugars and syngas using metabolically engineered Moorella thermoacetica
- PMID: 38281982
- PMCID: PMC10823632
- DOI: 10.1186/s13068-024-02460-1
Isopropanol production via the thermophilic bioconversion of sugars and syngas using metabolically engineered Moorella thermoacetica
Abstract
Background: Isopropanol (IPA) is a commodity chemical used as a solvent or raw material for polymeric products, such as plastics. Currently, IPA production depends largely on high-CO2-emission petrochemical methods that are not sustainable. Therefore, alternative low-CO2 emission methods are required. IPA bioproduction using biomass or waste gas is a promising method.
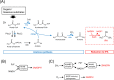
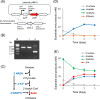
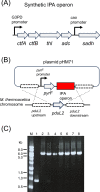
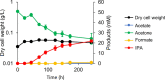
Results: Moorella thermoacetica, a thermophilic acetogenic microorganism, was genetically engineered to produce IPA. A metabolic pathway related to acetone reduction was selected, and acetone conversion to IPA was achieved via the heterologous expression of secondary alcohol dehydrogenase (sadh) in the thermophilic bacterium. sadh-expressing strains were combined with acetone-producing strains, to obtain an IPA-producing strain. The strain produced IPA as a major product using hexose and pentose sugars as substrates (81% mol-IPA/mol-sugar). Furthermore, IPA was produced from CO, whereas acetate was an abundant byproduct. Fermentation using syngas containing both CO and H2 resulted in higher IPA production at the specific rate of 0.03 h-1. The supply of reducing power for acetone conversion from the gaseous substrates was examined by supplementing acetone to the culture, and the continuous and rapid conversion of acetone to IPA showed a sufficient supply of NADPH for Sadh.
Conclusions: The successful engineering of M. thermoacetica resulted in high IPA production from sugars. M. thermoacetica metabolism showed a high capacity for acetone conversion to IPA in the gaseous substrates, indicating acetone production as the bottleneck in IPA production for further improving the strain. This study provides a platform for IPA production via the metabolic engineering of thermophilic acetogens.
Keywords: Biomass; Isopropanol production; Secondary alcohol dehydrogenase; Syngas; Thermophilic acetogen.
© 2024. The Author(s).
Conflict of interest statement
This work has been included in a patent application by Hiroshima University.
Figures


Similar articles
-
Genetic engineering of a thermophilic acetogen, Moorella thermoacetica Y72, to enable acetoin production.Front Bioeng Biotechnol. 2024 May 15;12:1398467. doi: 10.3389/fbioe.2024.1398467. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38812916 Free PMC article.
-
Progresses and challenges of engineering thermophilic acetogenic cell factories.Front Microbiol. 2024 Aug 30;15:1476253. doi: 10.3389/fmicb.2024.1476253. eCollection 2024. Front Microbiol. 2024. PMID: 39282569 Free PMC article. Review.
-
Metabolic engineering of Moorella thermoacetica for thermophilic bioconversion of gaseous substrates to a volatile chemical.AMB Express. 2021 Apr 23;11(1):59. doi: 10.1186/s13568-021-01220-w. AMB Express. 2021. PMID: 33891189 Free PMC article.
-
Thermodynamics and economic feasibility of acetone production from syngas using the thermophilic production host Moorella thermoacetica.Biotechnol Biofuels. 2017 Jun 12;10:150. doi: 10.1186/s13068-017-0827-8. eCollection 2017. Biotechnol Biofuels. 2017. PMID: 28616074 Free PMC article.
-
Thermophilic Moorella thermoacetica as a platform microorganism for C1 gas utilization: physiology, engineering, and applications.Bioresour Bioprocess. 2023 Sep 15;10(1):61. doi: 10.1186/s40643-023-00682-z. Bioresour Bioprocess. 2023. PMID: 38647965 Free PMC article. Review.
Cited by
-
Genetic engineering of a thermophilic acetogen, Moorella thermoacetica Y72, to enable acetoin production.Front Bioeng Biotechnol. 2024 May 15;12:1398467. doi: 10.3389/fbioe.2024.1398467. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38812916 Free PMC article.
-
Harnessing acetogenic bacteria for one-carbon valorization toward sustainable chemical production.RSC Chem Biol. 2024 Jul 8;5(9):812-832. doi: 10.1039/d4cb00099d. eCollection 2024 Aug 28. RSC Chem Biol. 2024. PMID: 39211478 Free PMC article. Review.
-
Isolation and characterization of novel acetogenic Moorella strains for employment as potential thermophilic biocatalysts.FEMS Microbiol Ecol. 2024 Aug 13;100(9):fiae109. doi: 10.1093/femsec/fiae109. FEMS Microbiol Ecol. 2024. PMID: 39118367 Free PMC article.
-
Progresses and challenges of engineering thermophilic acetogenic cell factories.Front Microbiol. 2024 Aug 30;15:1476253. doi: 10.3389/fmicb.2024.1476253. eCollection 2024. Front Microbiol. 2024. PMID: 39282569 Free PMC article. Review.
References
-
- Molino A, Chianese S, Musmarra D. Biomass gasification technology: the state of the art overview. J Energ Chem. 2016;25(1):10–25. doi: 10.1016/j.jechem.2015.11.005. - DOI
-
- Sikarwar VS, Zhao M, Clough P, Yao J, Zhong X, Memon MZ, et al. An overview of advances in biomass gasification. Energ Environ Sci. 2016;9(10):2939–2977. doi: 10.1039/C6EE00935B. - DOI
-
- Fackler N, Heijstra BD, Rasor BJ, Brown H, Martin J, Ni Z, et al. Stepping on the gas to a circular economy: accelerating development of carbon-negative chemical production from gas fermentation. Annu Rev Chem Biomol Eng. 2021;12:439–470. doi: 10.1146/annurev-chembioeng-120120-021122. - DOI - PubMed
-
- Wiegel J. Acetate and the potential of homoacetogenic bacteria for industrial applications. In: Drake HL, editor. Acetogenesis. New York: Springer; 1994.
Grants and funding
LinkOut - more resources
Full Text Sources
