Progesterone-induced blocking factor blockade causes hypertension in pregnant rats
- PMID: 38282604
- PMCID: PMC10832350
- DOI: 10.1111/aji.13805
Progesterone-induced blocking factor blockade causes hypertension in pregnant rats
Abstract
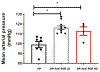
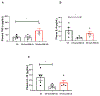
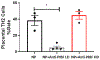
Preeclampsia (PE) is a multisystem disorder characterized by new onset hypertension in mid-late gestation and can include multi-organ dysfunction with or without proteinuria. It affects 5%-7% of all pregnancies in the U.S., making PE a major contributor to maternal and fetal morbidity and mortality. Currently, there is no cure for this pregnancy complication except for early delivery of the placenta and fetus. Moreover, the therapeutic options to treat PE are very limited. One potential trigger for the development of PE is progesterone deficiency-induced imbalance between T Helper 1(Th1)/Th2 cells, an increase in cytolytic natural killer (NK) cells and inflammatory cytokines that in turn leads to endothelial dysfunction, intrauterine growth restriction (IUGR) and hypertension. Importantly, progesterone signals the synthesis of progesterone-induced blocking factor (PIBF) which has anti-inflammatory effects and could promote the regulation of inflammation balance during pregnancy. However, the role of progesterone and PIBF in the pathophysiology of PE is still not fully understood. Thus, this current study was designed to test the hypothesis that inhibition of PIBF causes signs of PE in pregnant Sprague Dawley rats. In order to address our hypothesis, rabbit anti-PIBF IgG (0.25, low dose-LD or 0.50 mg/mL, high dose-HD) was administered intraperitoneally on gestation day (GD) 15 to normal pregnant Sprague Dawley (NP) rats. On GD 18, carotid catheters were inserted and on GD 19 mean blood pressure (MAP) and samples were collected for further analysis. MAP in normal pregnant rats (NP) rats (n = 7) was 99 ± 3 mmHg, which increased to 116 ± 2 mmHg in NP+ anti-PIBF LD (n = 10) and 113 ± 4 mmHg in NP+ anti-PIBF HD (n = 4), p <0 .05. Plasma TNF-alpha levels were 35 ± 8 pg/mL in NP rats and increased to 84 ± 21 pg/mL in NP+ Anti-PIBF HD (n = 4), p <0 .05. Plasma IL-4 and IL-10 levels were 22 ± 5 and 25+6 pg/mL in NP (n = 5), which decreased to 6 ± 1 and 8 ± 1 pg/mL in NP+ Anti-PIBF LD (n = 6, p < 0.05) and 16 ± 4 and 15 ± 5 pg/mL in NP+ Anti-PIBF HD (n = 4). Circulating total NK cells were 67 ± 11 % gate in NP rats (n = 3), which decreased to 28 ± 7% gate in NP+ Anti-PIBF LD and 45 ± 6% gate in NP+ Anti-PIBF HD. Cytolytic NK cells were increased in NP+ Anti-PIBF HD, p <0 .05. Moreover, circulating NO levels were significantly decreased while renal cortex PPET-1 levels increased NP+ Anti-PIBF HD. Our study demonstrates that PIBF blockade causes hypertension, inflammation and signs of endothelial dysfunction, all of which are associated with PE, thus indicating the importance of progesterone signalling pathways during a healthy pregnancy.
Keywords: inflammation; preeclampsia; pregnancy; progesterone-induced blocking factor.
© 2023 The Authors. American Journal of Reproductive Immunology published by John Wiley & Sons Ltd.
Conflict of interest statement
Figures



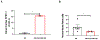
Similar articles
-
Progesterone-induced blocking factor improves blood pressure, inflammation, and pup weight in response to reduced uterine perfusion pressure (RUPP).Am J Physiol Regul Integr Comp Physiol. 2021 May 1;320(5):R719-R727. doi: 10.1152/ajpregu.00152.2020. Epub 2021 Feb 3. Am J Physiol Regul Integr Comp Physiol. 2021. PMID: 33533305 Free PMC article.
-
Progesterone Induced Blocking Factor Reduces Hypertension and Placental Mitochondrial Dysfunction in Response to sFlt-1 during Pregnancy.Cells. 2021 Oct 20;10(11):2817. doi: 10.3390/cells10112817. Cells. 2021. PMID: 34831040 Free PMC article.
-
Evaluation of maternal serum progesterone-induced blocking factor levels in pregnancies complicated with early- and late-onset preeclampsia.J Obstet Gynaecol. 2022 Aug;42(6):1991-1995. doi: 10.1080/01443615.2022.2056832. Epub 2022 Jun 1. J Obstet Gynaecol. 2022. PMID: 35648816
-
The Role of Extracellular Vesicles and PIBF in Embryo-Maternal Immune-Interactions.Front Immunol. 2018 Dec 13;9:2890. doi: 10.3389/fimmu.2018.02890. eCollection 2018. Front Immunol. 2018. PMID: 30619262 Free PMC article. Review.
-
PIBF: the double edged sword. Pregnancy and tumor.Am J Reprod Immunol. 2010 Aug 1;64(2):77-86. doi: 10.1111/j.1600-0897.2010.00833.x. Epub 2010 Mar 29. Am J Reprod Immunol. 2010. PMID: 20367622 Review.
References
-
- Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet Gynecol, 2013. 122(5): p. 1122–1131. - PubMed
-
- ACOG Practice Bulletin No. 202 Summary: Gestational Hypertension and Preeclampsia. Obstet Gynecol, 2019. 133(1): p. 1. - PubMed
-
- Hogan MC, et al., Maternal mortality for 181 countries, 1980–2008: a systematic analysis of progress towards Millennium Development Goal 5. Lancet, 2010. 375(9726): p. 1609–23. - PubMed
-
- Gilbert JS, et al., Pathophysiology of hypertension during preeclampsia: linking placental ischemia with endothelial dysfunction. Am J Physiol Heart Circ Physiol, 2008. 294(2): p. H541–50. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

