Visual guidance fine-tunes probing movements of an insect appendage
- PMID: 38285936
- PMCID: PMC10861887
- DOI: 10.1073/pnas.2306937121
Visual guidance fine-tunes probing movements of an insect appendage
Abstract
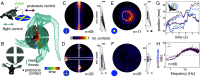
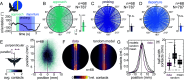
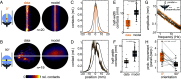
Visually guided reaching, a regular feature of human life, comprises an intricate neural control task. It includes identifying the target's position in 3D space, passing the representation to the motor system that controls the respective appendages, and adjusting ongoing movements using visual and proprioceptive feedback. Given the complexity of the neural control task, invertebrates, with their numerically constrained central nervous systems, are often considered incapable of this level of visuomotor guidance. Here, we provide mechanistic insights into visual appendage guidance in insects by studying the probing movements of the hummingbird hawkmoth's proboscis as they search for a flower's nectary. We show that visually guided proboscis movements fine-tune the coarse control provided by body movements in flight. By impairing the animals' view of their proboscis, we demonstrate that continuous visual feedback is required and actively sought out to guide this appendage. In doing so, we establish an insect model for the study of neural strategies underlying eye-appendage control in a simple nervous system.
Keywords: hawkmoth; insect; proboscis; visually guided reaching; visuomotor control.
Conflict of interest statement
Competing interests statement:The authors declare no competing interest.
Figures

Similar articles
-
The Hawkmoth Proboscis: An Insect Model for Sensorimotor Control of Reaching and Exploration.Integr Comp Biol. 2024 Nov 21;64(5):1354-1370. doi: 10.1093/icb/icae123. Integr Comp Biol. 2024. PMID: 39068501 Review.
-
The effect of visuomotor adaptation on proprioceptive localization: the contributions of perceptual and motor changes.Exp Brain Res. 2014 Jul;232(7):2073-86. doi: 10.1007/s00221-014-3896-y. Epub 2014 Mar 13. Exp Brain Res. 2014. PMID: 24623356
-
Visually-guided correction of hand reaching movements: The neurophysiological bases in the cerebral cortex.Vision Res. 2015 May;110(Pt B):244-56. doi: 10.1016/j.visres.2014.09.009. Epub 2014 Sep 28. Vision Res. 2015. PMID: 25264945 Review.
-
PET study of visually and non-visually guided finger movements in patients with severe pan-sensory neuropathies and healthy controls.Exp Brain Res. 1999 Oct;128(3):291-302. doi: 10.1007/s002210050849. Exp Brain Res. 1999. PMID: 10501801
-
Effect of visuomotor-map uncertainty on visuomotor adaptation.J Neurophysiol. 2012 Mar;107(6):1576-85. doi: 10.1152/jn.00204.2011. Epub 2011 Dec 21. J Neurophysiol. 2012. PMID: 22190631
References
-
- Georgopoulos A. P., Grillner S., Visuomotor coordination in reaching and locomotion. Science 245, 1209–1210 (1989). - PubMed
-
- Santamaria-Navarro A., Solà J., Andrade-Cetto J., Visual Guidance of Unmanned Aerial Manipulators (Springer, 2019).
-
- Goodale M. A., Transforming vision into action. Vision Res. 51, 1567–1587 (2011). - PubMed
-
- Pouget A., Snyder L. H., Computational approaches to sensorimotor transformations. Nat. Neurosci. 3, 1192–1198 (2000). - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

