PRDX6-iPLA2 aggravates neuroinflammation after ischemic stroke via regulating astrocytes-induced M1 microglia
- PMID: 38287382
- PMCID: PMC10823689
- DOI: 10.1186/s12964-024-01476-2
PRDX6-iPLA2 aggravates neuroinflammation after ischemic stroke via regulating astrocytes-induced M1 microglia
Abstract
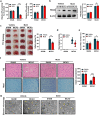
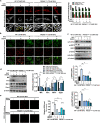
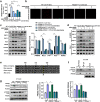
The crosstalk between astrocytes and microglia plays a pivotal role in neuroinflammation following ischemic stroke, and phenotypic distribution of these cells can change with the progression of ischemic stroke. Peroxiredoxin (PRDX) 6 phospholipase A2 (iPLA2) activity is involved in the generation of reactive oxygen species(ROS), with ROS driving the activation of microglia and astrocytes; however, its exact function remains unexplored. MJ33, PRDX6D140A mutation was used to block PRDX6-iPLA2 activity in vitro and vivo after ischemic stroke. PRDX6T177A mutation was used to block the phosphorylation of PRDX6 in CTX-TNA2 cell lines. NAC, GSK2795039, Mdivi-1, U0126, and SB202190 were used to block the activity of ROS, NOX2, mitochondrial fission, ERK, and P38, respectively, in CTX-TNA2 cells. In ischemic stroke, PRDX6 is mainly expressed in astrocytes and PRDX6-iPLA2 is involved in the activation of astrocytes and microglia. In co-culture system, Asp140 mutation in PRDX6 of CTX-TNA2 inhibited the polarization of microglia, reduced the production of ROS, suppressed NOX2 activation, and inhibited the Drp1-dependent mitochondrial fission following OGD/R. These effects were further strengthened by the inhibition of ROS production. In subsequent experiments, U0126 and SB202190 inhibited the phosphorylation of PRDX6 at Thr177 and reduced PRDX6-iPLA2 activity. These results suggest that PRDX6-iPLA2 plays an important role in the astrocyte-induced generation of ROS and activation of microglia, which are regulated by the activation of Nox2 and Drp1-dependent mitochondrial fission pathways. Additionally, PRDX6-iPLA2 activity is regulated by MAPKs via the phosphorylation of PRDX6 at Thr177 in astrocytes.
Keywords: Astrocytes; Drp1‑ dependent mitchondrial fission; Microglia; Nox2; PRDX6-iPLA2.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






Similar articles
-
Peroxiredoxin 6 phosphorylation and subsequent phospholipase A2 activity are required for agonist-mediated activation of NADPH oxidase in mouse pulmonary microvascular endothelium and alveolar macrophages.J Biol Chem. 2011 Apr 1;286(13):11696-706. doi: 10.1074/jbc.M110.206623. Epub 2011 Jan 24. J Biol Chem. 2011. PMID: 21262967 Free PMC article.
-
[Knockdown of PRDX6 in microglia reduces neuron viability after OGD/R injury].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2016 Aug;32(8):1014-20. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2016. PMID: 27412928 Chinese.
-
Phospholipase A2 of Peroxiredoxin 6 Plays a Critical Role in Cerebral Ischemia/Reperfusion Inflammatory Injury.Front Cell Neurosci. 2017 Apr 5;11:99. doi: 10.3389/fncel.2017.00099. eCollection 2017. Front Cell Neurosci. 2017. PMID: 28424593 Free PMC article.
-
The Relationship of Astrocytes and Microglia with Different Stages of Ischemic Stroke.Curr Neuropharmacol. 2023;21(12):2465-2480. doi: 10.2174/1570159X21666230718104634. Curr Neuropharmacol. 2023. PMID: 37464832 Free PMC article. Review.
-
Flavonoids and ischemic stroke-induced neuroinflammation: Focus on the glial cells.Biomed Pharmacother. 2024 Jan;170:115847. doi: 10.1016/j.biopha.2023.115847. Epub 2023 Nov 27. Biomed Pharmacother. 2024. PMID: 38016362 Review.
Cited by
-
Unveiling the Significance of Peroxiredoxin 6 in Central Nervous System Disorders.Antioxidants (Basel). 2024 Apr 10;13(4):449. doi: 10.3390/antiox13040449. Antioxidants (Basel). 2024. PMID: 38671897 Free PMC article. Review.
-
Short-Term Inhibition of NOX2 Prevents the Development of Aβ-Induced Pathology in Mice.Antioxidants (Basel). 2025 May 30;14(6):663. doi: 10.3390/antiox14060663. Antioxidants (Basel). 2025. PMID: 40563297 Free PMC article.
-
Peroxiredoxin 6 in Stress Orchestration and Disease Interplay.Antioxidants (Basel). 2025 Mar 23;14(4):379. doi: 10.3390/antiox14040379. Antioxidants (Basel). 2025. PMID: 40298631 Free PMC article. Review.
-
Therapeutic prospects and potential mechanisms of Prdx6: as a novel target in musculoskeletal disorders.Front Physiol. 2025 Apr 17;16:1524100. doi: 10.3389/fphys.2025.1524100. eCollection 2025. Front Physiol. 2025. PMID: 40313876 Free PMC article. Review.
-
Astrocyte-microglia crosstalk in subarachnoid hemorrhage: mechanisms and treatments.Front Immunol. 2025 Jun 30;16:1547858. doi: 10.3389/fimmu.2025.1547858. eCollection 2025. Front Immunol. 2025. PMID: 40661949 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

