Quinalizarin as a potential antifungal drug for the treatment of Candida albicans fungal infection in cancer patients
- PMID: 38289929
- PMCID: PMC10913734
- DOI: 10.1128/spectrum.03652-23
Quinalizarin as a potential antifungal drug for the treatment of Candida albicans fungal infection in cancer patients
Abstract
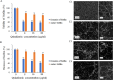
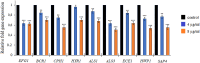
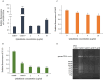
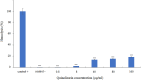
This study aims to analyze the antifungal properties of quinalizarin, a plant-derived compound with proven anticancer effects. Quinalizarin exhibited antifungal activity against opportunistic pathogenic Candida species and Geotrichum capitatum. The treatment with this anthraquinone reduced hyphal growth, inhibited biofilm formation, and damaged mature Candida albicans biofilms. Real-time RT-PCR revealed that quinalizarin downregulated the expression of hyphae-related and biofilm-specific genes. The flow cytometry method used in the study showed that both apoptosis and necrosis were the physiological mechanisms of quinalizarin-induced C. albicans cell death, depending on the dose of the antifungal agent. A further study revealed an increase in the levels of intracellular reactive oxygen species and alterations in mitochondrial membrane potential after treatment with quinalizarin. Finally, quinalizarin was found to have low toxicity in a hemolytic test using human erythrocytes. In conclusion, we have identified quinalizarin as a potential antifungal compound.IMPORTANCEThis article is a study to determine the antifungal activity of quinalizarin (1,2,5,8-tetrahydroxyanthraquinone). Quinalizarin has potential antitumor properties and is effective in different types of tumor cells. The aim of the present study was to prove that quinalizarin can be used simultaneously in the treatment of cancer and in the treatment of intercurrent fungal infections. Quinalizarin was identified as a novel antifungal compound with low toxicity. These results may contribute to the development of a new drug with dual activity in the treatment of cancer-associated candidiasis.
Keywords: Candida albicans; Candida auris; ROS; SEM; anthraquinones; antifungal activity; biofilm; cancer; fungal infection; hyphae; quinalizarin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources

