Stability indicating eco-friendly quantitation of terbutaline and its pro-drug bambuterol using quantitative proton nuclear magnetic spectroscopy
- PMID: 38291471
- PMCID: PMC10829239
- DOI: 10.1186/s13065-024-01120-7
Stability indicating eco-friendly quantitation of terbutaline and its pro-drug bambuterol using quantitative proton nuclear magnetic spectroscopy
Abstract
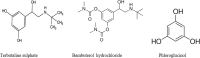
Quantitative 1H-NMR became an increasingly important issue in pharmaceutical analytical chemistry. This study used NMR spectroscopy to assay the bronchodilator drug terbutaline sulfate and its pro-drug bambuterol hydrochloride in pure form and pharmaceutical preparations. The technique proceeded using deuterium oxide (D2O) as an 1H-NMR solvent and phloroglucinol anhydrous as an internal standard (IS). Comparatively, to the phloroglucinol signal at 5.9 ppm, the resulting quantitative signals of the studied drugs were corrected. The terbutaline singlet signal at 6.3 ppm was chosen for quantification, while the bambuterol quantitative singlet signal was at 2.9 ppm. The two drugs were rectilinear over the concentration range of 1.0-16.0 mg/mL. LOD values were 0.19 and 0.21 mg/mL while LOQ values were 0.58 and 0.64 mg/mL for terbutaline and bambuterol respectively. The developed method has been validated according to the International Conference of Harmonization (ICH) regarding linearity, accuracy, precision, specificity, and robustness. A greenness profile assessment was applied, and the method proved to be green. The method enables the assay of the two drugs in pure drug and pharmaceutical preparations. The method also enables the assay of the two drugs in the presence of each other; thus, it is considered a stability-indicating method where terbutaline is an acid degradation product of bambuterol.
Keywords: Bambuterol; NMR spectroscopy; Terbutaline.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Svensson LA. Development of bambuterol; a long acting, orally active beta-agonist prodrug. Vienna: Springer; 1998.
-
- Commission BP, British Pharmacopoeia Her Majesty’s Stationary Office. 1998, London.
-
- Moreton RC. United States pharmacopeia-national formulary. J Excipients Food Chem. 2016;6(3):925.
LinkOut - more resources
Full Text Sources