Tandem Hock and Friedel-Crafts reactions allowing an expedient synthesis of a cyclolignan-type scaffold
- PMID: 38292045
- PMCID: PMC10825801
- DOI: 10.3762/bjoc.20.15
Tandem Hock and Friedel-Crafts reactions allowing an expedient synthesis of a cyclolignan-type scaffold
Abstract
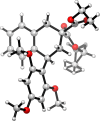
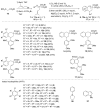
The Hock cleavage, which is compatible with tandem processes, was applied to the synthesis of 1-aryltetralines through a one-pot transformation from readily available benzyl(prenyl)malonate substrates. After the photooxygenation of the prenyl moiety, the resulting hydroperoxide was directly engaged in a Hock cleavage by adding a Lewis acid. The presence of an aromatic nucleophile in the reaction mixture and that of a benzyl moiety on the substrate resulted in tandem Friedel-Crafts reactions to form the 1-aryltetraline products. These compounds share a close analogy to the cyclolignan natural products. Experimental observations and a DFT study support the involvement of an aldehyde intermediate during the Friedel-Crafts reactions, rather than an oxocarbenium.
Keywords: 1-aryltetralines; Friedel–Crafts reaction; Hock rearrangement; oxidative cleavage; tandem reactions.
Copyright © 2024, Ikonnikova et al.
Figures





References
-
- Hock H, Lang S. Ber Dtsch Chem Ges B. 1944;77:257–264. doi: 10.1002/cber.19440770321. - DOI
-
- Weber M, Weber M, Weber V. Ullmann’s Encyclopedia of Industrial Chemistry. Weinheim, Germany: Wiley-VCH; 2020. Phenol; pp. 1–20. - DOI
-
- Turconi J, Griolet F, Guevel R, Oddon G, Villa R, Geatti A, Hvala M, Rossen K, Göller R, Burgard A. Org Process Res Dev. 2014;18:417–422. doi: 10.1021/op4003196. - DOI
-
- Corey E J, Nagata R, Wright S W. Tetrahedron Lett. 1987;28:4917–4920. doi: 10.1016/s0040-4039(00)96658-9. - DOI
-
- Sy L-K, Brown G D. Tetrahedron. 2002;58:897–908. doi: 10.1016/s0040-4020(01)01193-0. - DOI
LinkOut - more resources
Full Text Sources
