Impaired value-based decision-making in Parkinson's disease apathy
- PMID: 38305691
- PMCID: PMC10994558
- DOI: 10.1093/brain/awae025
Impaired value-based decision-making in Parkinson's disease apathy
Abstract
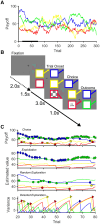
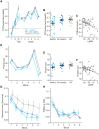
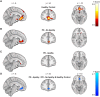
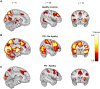
Apathy is a common and disabling complication of Parkinson's disease characterized by reduced goal-directed behaviour. Several studies have reported dysfunction within prefrontal cortical regions and projections from brainstem nuclei whose neuromodulators include dopamine, serotonin and noradrenaline. Work in animal and human neuroscience have confirmed contributions of these neuromodulators on aspects of motivated decision-making. Specifically, these neuromodulators have overlapping contributions to encoding the value of decisions, and influence whether to explore alternative courses of action or persist in an existing strategy to achieve a rewarding goal. Building upon this work, we hypothesized that apathy in Parkinson's disease should be associated with an impairment in value-based learning. Using a four-armed restless bandit reinforcement learning task, we studied decision-making in 75 volunteers; 53 patients with Parkinson's disease, with and without clinical apathy, and 22 age-matched healthy control subjects. Patients with apathy exhibited impaired ability to choose the highest value bandit. Task performance predicted an individual patient's apathy severity measured using the Lille Apathy Rating Scale (R = -0.46, P < 0.001). Computational modelling of the patient's choices confirmed the apathy group made decisions that were indifferent to the learnt value of the options, consistent with previous reports of reward insensitivity. Further analysis demonstrated a shift away from exploiting the highest value option and a reduction in perseveration, which also correlated with apathy scores (R = -0.5, P < 0.001). We went on to acquire functional MRI in 59 volunteers; a group of 19 patients with and 20 without apathy and 20 age-matched controls performing the Restless Bandit Task. Analysis of the functional MRI signal at the point of reward feedback confirmed diminished signal within ventromedial prefrontal cortex in Parkinson's disease, which was more marked in apathy, but not predictive of their individual apathy severity. Using a model-based categorization of choice type, decisions to explore lower value bandits in the apathy group activated prefrontal cortex to a similar degree to the age-matched controls. In contrast, Parkinson's patients without apathy demonstrated significantly increased activation across a distributed thalamo-cortical network. Enhanced activity in the thalamus predicted individual apathy severity across both patient groups and exhibited functional connectivity with dorsal anterior cingulate cortex and anterior insula. Given that task performance in patients without apathy was no different to the age-matched control subjects, we interpret the recruitment of this network as a possible compensatory mechanism, which compensates against symptomatic manifestation of apathy in Parkinson's disease.
Keywords: Parkinson’s disease; apathy; computational modelling; decision-making; functional MRI; reward insensitivity.
© The Author(s) 2024. Published by Oxford University Press on behalf of the Guarantors of Brain.
Conflict of interest statement
The authors report no competing interests.
Figures


Comment in
-
Learning the value of experience.Brain. 2024 Apr 4;147(4):1127-1129. doi: 10.1093/brain/awae084. Brain. 2024. PMID: 38530634 No abstract available.
References
-
- Starkstein SE, Mayberg HS, Preziosi T, Andrezejewski P, Leiguarda R, Robinson R. Reliability, validity, and clinical correlates of apathy in Parkinson’s disease. J Neuropsychiatry Clin Neurosci. 1992;4:134–139. - PubMed
-
- den Brok MG, van Dalen JW, van Gool WA, Moll van Charante EP, de Bie RM, Richard E. Apathy in Parkinson's disease: A systematic review and meta-analysis. Mov Disord. 2015;30:759–769. - PubMed
-
- Pedersen KF, Larsen JP, Alves G, Aarsland D. Prevalence and clinical correlates of apathy in Parkinson's disease: A community-based study. Parkinsonism Relat Disord. 2009;15:295–299. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

