Dynamic tracking of human umbilical cord mesenchymal stem cells (hUC-MSCs) following intravenous administration in mice model
- PMID: 38314402
- PMCID: PMC10834363
- DOI: 10.1016/j.reth.2024.01.003
Dynamic tracking of human umbilical cord mesenchymal stem cells (hUC-MSCs) following intravenous administration in mice model
Abstract
Introduction: In the past decades, human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) have sparked interest in cellular therapy due to their immunomodulatory properties. Nevertheless, the fate of hUC-MSCs in the body remains poorly understood. This study aimed to investigate the biodistribution, homing and clearance of systemically administered hUC-MSCs in healthy BALB/c mice model.
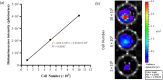
Methods: hUC-MSCs were labelled with GFP-Luc2 protein, followed by characterisation with flow cytometry. Upon intravenous infusion of transduced hUC-MSCs into the healthy BALB/c mice, the cells were dynamically monitored through the bioluminescent imaging (BLI) approach.
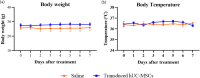
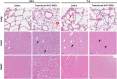
Results: Transduction of hUC-MSCs with GFP-Luc2 not only preserved the characteristics of MSCs, but also allowed live monitoring of transduced cells in the mice model. Upon systemic administration, BLI showed that transduced hUC-MSCs first localised predominantly in the lungs of healthy BALB/c mice and mainly remained in the lungs for up to 3 days before eventually cleared from the body. At terminal sacrifice, plasma chemistry biomarkers remained unchanged except for C-peptide levels, which were significantly reduced in the hUC-MSCs group. Histopathological findings further revealed that hUC-MSCs infusion did not cause any adverse effects and toxicity to lung, liver and heart tissues.
Conclusions: Collectively, systemically administrated hUC-MSCs was safe and demonstrated dynamic homing capacity before eventually disappearing from the body.
Keywords: Biodistribution; GFP-Luc2; Human umbilical cord mesenchymal stem cell.
© 2024 The Japanese Society for Regenerative Medicine. Production and hosting by Elsevier B.V.
Conflict of interest statement
SP Chin advices Cytopeutics on regulatory, clinical and research activities. Information pertaining to writing assistance: No funded writing assistance was utilized in the production of this manuscript.
Figures




Similar articles
-
[SOX9 enhanced chondrogenic differentiation potential of human umbilical cord mesenchymal stem cells through cellular aggregation].Zhonghua Yi Xue Za Zhi. 2012 Aug 7;92(29):2050-4. Zhonghua Yi Xue Za Zhi. 2012. PMID: 23253807 Chinese.
-
Preclinical assessments of safety and tumorigenicity of very high doses of allogeneic human umbilical cord mesenchymal stem cells.In Vitro Cell Dev Biol Anim. 2024 Mar;60(3):307-319. doi: 10.1007/s11626-024-00852-z. Epub 2024 Feb 29. In Vitro Cell Dev Biol Anim. 2024. PMID: 38421574 Free PMC article.
-
[Effects of human mesenchymal stem cells on airway inflammation in allergic asthma mice and the underlying mechanism].Zhonghua Yi Xue Za Zhi. 2017 Sep 12;97(34):2697-2702. doi: 10.3760/cma.j.issn.0376-2491.2017.34.012. Zhonghua Yi Xue Za Zhi. 2017. PMID: 28910960 Chinese.
-
What is the impact of human umbilical cord mesenchymal stem cell transplantation on clinical treatment?Stem Cell Res Ther. 2020 Dec 1;11(1):519. doi: 10.1186/s13287-020-02011-z. Stem Cell Res Ther. 2020. PMID: 33261658 Free PMC article. Review.
-
Human Umbilical Cord Mesenchymal Stem Cells' Cultivation and Treatment of Liver Diseases.Curr Stem Cell Res Ther. 2023;18(3):286-298. doi: 10.2174/1574888X17666220623111406. Curr Stem Cell Res Ther. 2023. PMID: 35747963 Review.
Cited by
-
Therapeutic potential of mesenchymal stem cells for fungal infections: mechanisms, applications, and challenges.Front Microbiol. 2025 Jan 30;16:1554917. doi: 10.3389/fmicb.2025.1554917. eCollection 2025. Front Microbiol. 2025. PMID: 39949625 Free PMC article. Review.
-
Genetically Modified Mesenchymal Stromal/Stem Cells as a Delivery Platform for SE-33, a Cathelicidin LL-37 Analogue: Preclinical Pharmacokinetics and Tissue Distribution in C57BL/6 Mice.Antibiotics (Basel). 2025 Apr 24;14(5):429. doi: 10.3390/antibiotics14050429. Antibiotics (Basel). 2025. PMID: 40426496 Free PMC article.
-
Preclinical Evaluation of the Safety, Toxicity and Efficacy of Genetically Modified Wharton's Jelly Mesenchymal Stem/Stromal Cells Expressing the Antimicrobial Peptide SE-33.Cells. 2025 Feb 26;14(5):341. doi: 10.3390/cells14050341. Cells. 2025. PMID: 40072070 Free PMC article.
-
Rebuilding the myocardial microenvironment to enhance mesenchymal stem cells-mediated regeneration in ischemic heart disease.Front Bioeng Biotechnol. 2024 Sep 20;12:1468833. doi: 10.3389/fbioe.2024.1468833. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39372432 Free PMC article. Review.
References
-
- Costa L.A., Eiro N., Fraile M., Gonzalez L.O., Saá J., Garcia-Portabella P., et al. Functional heterogeneity of mesenchymal stem cells from natural niches to culture conditions: implications for further clinical uses. Cell Mol Life Sci. 2021;78:447–467. doi: 10.1007/S00018-020-03600-0/TABLES/2. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

