A high-throughput target-based screening approach for the identification and assessment of Mycobacterium tuberculosis mycothione reductase inhibitors
- PMID: 38315026
- PMCID: PMC10913476
- DOI: 10.1128/spectrum.03723-23
A high-throughput target-based screening approach for the identification and assessment of Mycobacterium tuberculosis mycothione reductase inhibitors
Abstract
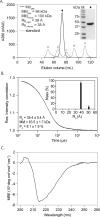
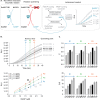
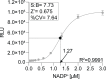
The World Health Organization's goal to combat tuberculosis (TB) is hindered by the emergence of anti-microbial resistance, therefore necessitating the exploration of new drug targets. Multidrug regimens are indispensable in TB therapy as they provide synergetic bactericidal effects, shorten treatment duration, and reduce the risk of resistance development. The research within our European RespiriTB consortium explores Mycobacterium tuberculosis energy metabolism to identify new drug candidates that synergize with bedaquiline, with the aim of discovering more efficient combination drug regimens. In this study, we describe the development and validation of a luminescence-coupled, target-based assay for the identification of novel compounds inhibiting Mycobacterium tuberculosis mycothione reductase (MtrMtb), an enzyme with a role in the protection against oxidative stress. Recombinant MtrMtb was employed for the development of a highly sensitive, robust high-throughput screening (HTS) assay by coupling enzyme activity to a bioluminescent readout. Its application in a semi-automated setting resulted in the screening of a diverse library of ~130,000 compounds, from which 19 hits were retained after an assessment of their potency, selectivity, and specificity. The selected hits formed two clusters and four fragment molecules, which were further evaluated in whole-cell and intracellular infection assays. The established HTS discovery pipeline offers an opportunity to deliver novel MtrMtb inhibitors and lays the foundation for future efforts in developing robust biochemical assays for the identification and triaging of inhibitors from high-throughput library screens.
Importance: The growing anti-microbial resistance poses a global public health threat, impeding progress toward eradicating tuberculosis. Despite decades of active research, there is still a dire need for the discovery of drugs with novel modes of action and exploration of combination drug regimens. Within the European RespiriTB consortium, we explore Mycobacterium tuberculosis energy metabolism to identify new drug candidates that synergize with bedaquiline, with the aim of discovering more efficient combination drug regimens. In this study, we present the development of a high-throughput screening pipeline that led to the identification of M. tuberculosis mycothione reductase inhibitors.
Keywords: drug discovery; high-throughput screening; mycothione reductase; tuberculosis.
Conflict of interest statement
K.T. and D.L. are employees of Janssen Pharmaceutica. N.V., N.S., L.O., K.V.C., L.D.V., R.G., H.P.M., K.A., S.V.S., D.C., Y.G.-J.S., and P.C. declare no conflicts of interest.
Figures


Similar articles
-
Target Discovery for New Antitubercular Drugs Using a Large Dataset of Growth Inhibitors from PubChem.Infect Disord Drug Targets. 2020;20(3):352-366. doi: 10.2174/1871526519666181205163810. Infect Disord Drug Targets. 2020. PMID: 30520384 Review.
-
Strategies to Combat Multi-Drug Resistance in Tuberculosis.Acc Chem Res. 2021 May 18;54(10):2361-2376. doi: 10.1021/acs.accounts.0c00878. Epub 2021 Apr 22. Acc Chem Res. 2021. PMID: 33886255 Free PMC article. Review.
-
Identification of Chemical Scaffolds Targeting Drug-Resistant and Latent Mycobacterium tuberculosis through High-Throughput Whole-Cell Screening.ACS Infect Dis. 2024 Feb 9;10(2):513-526. doi: 10.1021/acsinfecdis.3c00463. Epub 2024 Jan 18. ACS Infect Dis. 2024. PMID: 38238154
-
[Development of antituberculous drugs: current status and future prospects].Kekkaku. 2006 Dec;81(12):753-74. Kekkaku. 2006. PMID: 17240921 Review. Japanese.
-
[Recent progress in mycobacteriology].Kekkaku. 2007 Oct;82(10):783-99. Kekkaku. 2007. PMID: 18018602 Japanese.
Cited by
-
Breaking the energy chain: importance of ATP synthase in Mycobacterium tuberculosis and its potential as a drug target.RSC Med Chem. 2025 Jan 8. doi: 10.1039/d4md00829d. Online ahead of print. RSC Med Chem. 2025. PMID: 39790127 Free PMC article. Review.
-
A new paradigm for drug discovery in the treatment of complex diseases: drug discovery and optimization.Chin Med. 2025 Mar 24;20(1):40. doi: 10.1186/s13020-025-01075-4. Chin Med. 2025. PMID: 40122800 Free PMC article. Review.
-
Novel Prodrug Strategies for the Treatment of Tuberculosis.Chem Asian J. 2024 Dec 2;19(23):e202400944. doi: 10.1002/asia.202400944. Epub 2024 Oct 24. Chem Asian J. 2024. PMID: 39179514 Free PMC article. Review.
References
-
- World Health Organisation . 2022. Global tuberculosis report 2022. Available from: https://www.who.int/publications-detail-redirect/9789240061729
-
- World Health Organisation . 2020. Guidance for the surveillance of drug resistance in tuberculosis. 6th ed. World Health Organization.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

