A GIPR antagonist conjugated to GLP-1 analogues promotes weight loss with improved metabolic parameters in preclinical and phase 1 settings
- PMID: 38316982
- PMCID: PMC10896721
- DOI: 10.1038/s42255-023-00966-w
A GIPR antagonist conjugated to GLP-1 analogues promotes weight loss with improved metabolic parameters in preclinical and phase 1 settings
Abstract
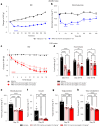
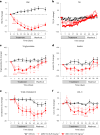
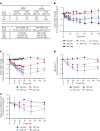
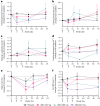
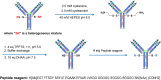
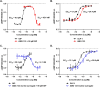
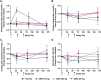
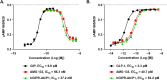
Obesity is a major public health crisis. Multi-specific peptides have emerged as promising therapeutic strategies for clinical weight loss. Glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are endogenous incretins that regulate weight through their receptors (R). AMG 133 (maridebart cafraglutide) is a bispecific molecule engineered by conjugating a fully human monoclonal anti-human GIPR antagonist antibody to two GLP-1 analogue agonist peptides using amino acid linkers. Here, we confirm the GIPR antagonist and GLP-1R agonist activities in cell-based systems and report the ability of AMG 133 to reduce body weight and improve metabolic markers in male obese mice and cynomolgus monkeys. In a phase 1, randomized, double-blind, placebo-controlled clinical study in participants with obesity ( NCT04478708 ), AMG 133 had an acceptable safety and tolerability profile along with pronounced dose-dependent weight loss. In the multiple ascending dose cohorts, weight loss was maintained for up to 150 days after the last dose. These findings support continued clinical evaluation of AMG 133.
© 2024. The Author(s).
Conflict of interest statement
M.M.V., S.C.L., L.A., R.K., S.S., Y.C., B.W., J.R.F., T.H., V.A.T., M.A., L.S., Y.Z., V.K., R.J., R.K.O., J.R.P., N.H. and J.L.S. are or were previously employees and stockholders of Amgen. J.N. declares no competing interests.
Figures

References
-
- World Health Organization. Obesity and Overweighthttps://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight (2021).
-
- Apovian, C. M. Obesity: definition, comorbidities, causes, and burden. Am. J. Manag Care22, s176–s185 (2016). - PubMed
-
- CDC. Adult Obesity Factshttps://www.cdc.gov/obesity/data/adult.html (2022).
-
- Wright, S. M. & Aronne, L. J. Obesity in 2010: the future of obesity medicine: where do we go from here? Nat. Rev. Endocrinol.7, 69–70 (2011). - PubMed
-
- Valsamakis, G., Konstantakou, P. & Mastorakos, G. New targets for drug treatment of obesity. Annu. Rev. Pharmacol. Toxicol.57, 585–605 (2017). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

