ROS-induced oxidative stress and mitochondrial dysfunction: a possible mechanism responsible for noise-induced ribbon synaptic damage
- PMID: 38322575
- PMCID: PMC10839402
- DOI: 10.62347/EVDE9449
ROS-induced oxidative stress and mitochondrial dysfunction: a possible mechanism responsible for noise-induced ribbon synaptic damage
Abstract
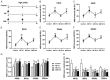
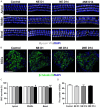
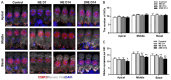
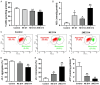
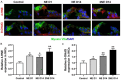
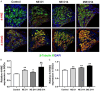
Evidence suggests that damage to the ribbon synapses (RS) may be the main cause of auditory dysfunction in noise-induced hearing loss (NIHL). Oxidative stress is implicated in the pathophysiology of synaptic damage. However, the relationship between oxidative stress and RS damage in NIHL remains unclear. To investigate the hypothesis that noise-induced oxidative stress is a key factor in synaptic damage within the inner ear, we conducted a study using mice subjected to single or repeated noise exposure (NE). We assessed auditory function using auditory brainstem response (ABR) test and examined cochlear morphology by immunofluorescence staining. The results showed that mice that experienced a single NE exhibited a threshold shift and recovered within two weeks. The ABR wave I latencies were prolonged, and the amplitudes decreased, suggesting RS dysfunction. These changes were also demonstrated by the loss of RS as evidenced by immunofluorescence staining. However, we observed threshold shifts that did not return to baseline levels following secondary NE. Additionally, ABR wave I latencies and amplitudes exhibited notable changes. Immunofluorescence staining indicated not only severe damage to RS but also loss of outer hair cells. We also noted decreased T-AOC, ATP, and mitochondrial membrane potential levels, alongside increased hydrogen peroxide concentrations post-NE. Furthermore, the expression levels of 4-HNE and 8-OHdG in the cochlea were notably elevated. Collectively, our findings suggest that the production of reactive oxygen species leads to oxidative damage in the cochlea. This mitochondrial dysfunction consequently contributes to the loss of RS, precipitating an early onset of NIHL.
Keywords: Noise-induced hearing loss; mitochondrial dysfunction; oxidative stress; reactive oxygen species; ribbon synapses.
AJTR Copyright © 2024.
Conflict of interest statement
None.
Figures
Similar articles
-
Dose-Dependent Pattern of Cochlear Synaptic Degeneration in C57BL/6J Mice Induced by Repeated Noise Exposure.Neural Plast. 2021 Jun 9;2021:9919977. doi: 10.1155/2021/9919977. eCollection 2021. Neural Plast. 2021. PMID: 34221004 Free PMC article.
-
Killer or helper? The mechanism underlying the role of adenylate activated kinase in sound conditioning.Front Synaptic Neurosci. 2022 Sep 7;14:940788. doi: 10.3389/fnsyn.2022.940788. eCollection 2022. Front Synaptic Neurosci. 2022. PMID: 36160917 Free PMC article.
-
Long-Term Conductive Auditory Deprivation During Early Development Causes Irreversible Hearing Impairment and Cochlear Synaptic Disruption.Neuroscience. 2019 May 15;406:345-355. doi: 10.1016/j.neuroscience.2019.01.065. Epub 2019 Feb 8. Neuroscience. 2019. PMID: 30742960
-
Candidate's thesis: enhancing intrinsic cochlear stress defenses to reduce noise-induced hearing loss.Laryngoscope. 2002 Sep;112(9):1515-32. doi: 10.1097/00005537-200209000-00001. Laryngoscope. 2002. PMID: 12352659
-
mito-TEMPO Attenuates Oxidative Stress and Mitochondrial Dysfunction in Noise-Induced Hearing Loss via Maintaining TFAM-mtDNA Interaction and Mitochondrial Biogenesis.Front Cell Neurosci. 2022 Feb 8;16:803718. doi: 10.3389/fncel.2022.803718. eCollection 2022. Front Cell Neurosci. 2022. PMID: 35210991 Free PMC article.
Cited by
-
Drug-Therapeutic Strategies for Noise-Induced Hearing Loss: A Narrative Review.Noise Health. 2025 May-Jun 01;27(126):203-209. doi: 10.4103/nah.nah_187_24. Epub 2025 Jun 26. Noise Health. 2025. PMID: 40574290 Free PMC article. Review.
-
Strategic disruption of cancer's powerhouse: precise nanomedicine targeting of mitochondrial metabolism.J Nanobiotechnology. 2024 Jun 8;22(1):318. doi: 10.1186/s12951-024-02585-3. J Nanobiotechnology. 2024. PMID: 38849914 Free PMC article. Review.
-
A Consolidated Understanding of the Contribution of Redox Dysregulation in the Development of Hearing Impairment.Antioxidants (Basel). 2024 May 13;13(5):598. doi: 10.3390/antiox13050598. Antioxidants (Basel). 2024. PMID: 38790703 Free PMC article. Review.
-
Synergistic Inhibition of Colon Cancer Cell Proliferation via p53, Bax, and Bcl-2 Modulation by Curcumin and Plumbagin Combination.ACS Omega. 2025 Apr 29;10(18):19045-19060. doi: 10.1021/acsomega.5c01258. eCollection 2025 May 13. ACS Omega. 2025. PMID: 40385152 Free PMC article.
-
Exploring biomarkers for noise-induced hearing loss through mitochondrial DNA methylation analysis.Front Pharmacol. 2025 Jul 4;16:1561791. doi: 10.3389/fphar.2025.1561791. eCollection 2025. Front Pharmacol. 2025. PMID: 40689209 Free PMC article.
References
-
- Cui Y, Sun GW, Yamashita D, Kanzaki S, Matsunaga T, Fujii M, Kaga K, Ogawa K. Acoustic overstimulation-induced apoptosis in fibrocytes of the cochlear spiral limbus of mice. Eur Arch Otorhinolaryngol. 2011;268:973–978. - PubMed
LinkOut - more resources
Full Text Sources
