Emerging roles for the ADAMTS-like family of matricellular proteins in cardiovascular disease through regulation of the extracellular microenvironment
- PMID: 38324186
- PMCID: PMC10850197
- DOI: 10.1007/s11033-024-09255-5
Emerging roles for the ADAMTS-like family of matricellular proteins in cardiovascular disease through regulation of the extracellular microenvironment
Abstract
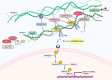
Dysregulation of the extracellular matrix (ECM) occurs widely across cardiovascular pathologies. Recent work has revealed important roles for the «a disintegrin-like and metalloprotease domain with thrombospondin-type 1 motifs like" (ADAMTSL) family of secreted glycoproteins in cardiovascular tissues during development and disease. Key insights in this regard have come from naturally occurring gene mutations in humans and animals that result in severe diseases with cardiovascular manifestations or aortopathies. Expression of ADAMTSL genes is greatly increased in the myocardium during heart failure. Genetically modified mice recapitulate phenotypes of patients with ADAMTSL mutations and demonstrate important functions in the ECM. The novel functions thus disclosed are intriguing because, while these proteins are neither structural, nor proteases like the related ADAMTS proteases, they appear to act as regulatory, i.e., matricellular proteins. Evidence from genetic variants, genetically engineered mouse mutants, and in vitro investigations have revealed regulatory functions of ADAMTSLs related to fibrillin microfibrils and growth factor signaling. Interestingly, the ability to regulate transforming growth factor (TGF)β signaling may be a shared characteristic of some ADAMTSLs. TGFβ signaling is important in cardiovascular development, health and disease and a central driver of ECM remodeling and cardiac fibrosis. New strategies to target dysregulated TGFβ signaling are warranted in aortopathies and cardiac fibrosis. With their emerging roles in cardiovascular tissues, the ADAMTSL proteins may provide causative genes, diagnostic biomarkers and novel treatment targets in cardiovascular disease. Here, we discuss the relevance of ADAMTSLs to cardiovascular medicine.
Keywords: Aortic aneurysms; Cardiac fibrosis; Extracellular matrix; Growth factor signaling; Heart failure; Microfibrils.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Secreted ADAMTS-like proteins as regulators of connective tissue function.Am J Physiol Cell Physiol. 2024 Mar 1;326(3):C756-C767. doi: 10.1152/ajpcell.00680.2023. Epub 2024 Jan 29. Am J Physiol Cell Physiol. 2024. PMID: 38284126 Review.
-
The extracellular matrix glycoprotein ADAMTSL2 is increased in heart failure and inhibits TGFβ signalling in cardiac fibroblasts.Sci Rep. 2021 Oct 5;11(1):19757. doi: 10.1038/s41598-021-99032-2. Sci Rep. 2021. PMID: 34611183 Free PMC article.
-
Basic Components of Connective Tissues and Extracellular Matrix: Fibronectin, Fibrinogen, Laminin, Elastin, Fibrillins, Fibulins, Matrilins, Tenascins and Thrombospondins.Adv Exp Med Biol. 2021;1348:105-126. doi: 10.1007/978-3-030-80614-9_4. Adv Exp Med Biol. 2021. PMID: 34807416
-
Inhibition of the extracellular enzyme A disintegrin and metalloprotease with thrombospondin motif 4 prevents cardiac fibrosis and dysfunction.Cardiovasc Res. 2023 Aug 19;119(10):1915-1927. doi: 10.1093/cvr/cvad078. Cardiovasc Res. 2023. PMID: 37216909 Free PMC article.
-
ADAMTS proteins as modulators of microfibril formation and function.Matrix Biol. 2015 Sep;47:34-43. doi: 10.1016/j.matbio.2015.05.004. Epub 2015 May 7. Matrix Biol. 2015. PMID: 25957949 Free PMC article. Review.
Cited by
-
Unraveling the Role of RSPRY1 in TGF-β Pathway Dysregulation: Insights into the Pathogenesis of Spondyloepimetaphyseal Dysplasia.Int J Mol Sci. 2025 Jan 28;26(3):1134. doi: 10.3390/ijms26031134. Int J Mol Sci. 2025. PMID: 39940902 Free PMC article.
-
Modulation of metabolic, inflammatory and fibrotic pathways by semaglutide in metabolic dysfunction-associated steatohepatitis.Nat Med. 2025 Jul 21. doi: 10.1038/s41591-025-03799-0. Online ahead of print. Nat Med. 2025. PMID: 40691365
References
-
- Tsao CW et al (2022) Heart Disease and Stroke Statistics-2022 update: a Report from the American Heart Association. Circulation 145(8):e153–e639 - PubMed
-
- Díez J, de Boer RA (2022) Management of cardiac fibrosis is the largest unmet medical need in heart failure. Cardiovasc Res 118(2):e20–e22 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

