Converging survival trends in non-small cell lung cancer patients with and without brain metastasis receiving state-of-the-art treatment
- PMID: 38324192
- PMCID: PMC10876498
- DOI: 10.1007/s11060-024-04562-0
Converging survival trends in non-small cell lung cancer patients with and without brain metastasis receiving state-of-the-art treatment
Abstract
Introduction: Historically, patients with brain metastasis (BM) have been excluded from clinical trials investigating treatments for non-small cell lung cancer (NSCLC) due to their unfavorable prognosis. Advanced treatments have increased survival prospects for NSCLC patients with BM. This study evaluated the life expectancy of NSCLC patients with and without BM in the context of contemporary treatments.
Methods: Outcome data were collected for patients with advanced NSCLC attending a tertiary medical center between 2015 and 2020. Patients were stratified according to BM status and compared for overall survival (OS) using log-rank and Cox regression analyses.
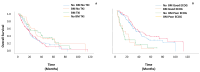
Results: The cohort included 360 patients with NSCLC of whom 134 (37.2%) had BM. Most (95%) of cases of BM developed within the first two years: 63% at diagnosis, 18% during the first year, 14% during the second year. There was no significant difference in OS between patients without BM and those with BM (median 23.7 vs. 22.3 months, HR = 0.97, p = 0.82); patients with BM and a targetable or non-targetable mutation (40.2 vs. 31.4 months, HR = 0.93, p = 0.84, and 20.7 vs. 19.87 months, HR = 0.95, p = 0.75, respectively); and patients with symptomatic BM (23.7 vs. 19.8 months, HR = 0.95, p = 0.78). Treatment for BM (95% of patients) consisted of stereotactic radiosurgery or tyrosine kinase inhibitors, with corresponding intracranial control rates of 90% and 86%.
Conclusion: The results imply that the presence of BM has no impact on the prognosis of NSCLC. The practice of excluding NSCLC patients with BM from clinical trials warrants reconsideration.
Keywords: Brain metastasis; Non small cell lung cancer; Prognosis.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Itamar Averbuch reports receiving personal fees from Merck Serono outside the submitted work. Alona Zer reports receiving consulting fees from Nixio; payment/honoraria from MSD, Takeda, and AstraZeneca; support for meetings/travel from MSD; participation on Data Safety Monitoring or Advisory Boards for XAIR and AstraZeneca; stock/stock options in Nixio, outside the submitted work. Mor Moskovitz reports receiving grants/contracts from Rabin, Southwestern University, and AstraZeneca; consulting fees from MSD, AstraZeneca, Takeda, Pfizer, Roche, Novartis; payment/honoraria from MSD, AstraZeneca, Takeda, Pfizer, Roche, BMS, JNJ; support for meetings/travel from Pfizer, Roche, outside the submitted work. Oded Icht reports receiving consulting fees from Brainlab; payment/honoraria from AstraZeneca and Roche, outside the submitted work. Ofer Rotem reports receiving consulting fees from AstraZeneca, Boehringer Ingelheim, BMS, Edocate, MSD, Novartis, Nucleai, Pfizer, Rhenium, Roche, Takeda, Teva; payment/honoraria and support for meetings/travel from AstraZeneca, Boehringer Ingelheim, BMS, MSD, Novartis, Pfizer, Rhenium, Roche, Takeda, Teva, outside the submitted work. Daniel Reinhorn reports receiving payment/honoraria from BMS and AstraZeneca, outside the submitted work. Shlomit Yust-Katz reports receiving grants/contracts from MSK; consulting fees from Pfizer; payment/honoraria from Pfizer, Teva, Eli-Lilly, and Abbvie; support for meetings/travel from Novocure; participation on Data Safety Monitoring or Advisory Boards from Pfizer, outside the submitted work. Roi Tschernichovsky, Dror Limon, Noga Kurman, Ekaterina Hanovich, Alexandra Benouaich-Amiel, Tali Siegal, and Omer Gal report no conflict of interest.
Figures




Similar articles
-
Targeted Therapies and Utility of the Lung-molGPA in Non-Small-Cell Lung Cancer Patients with Brain Metastases.Oncology. 2022;100(10):542-554. doi: 10.1159/000525082. Epub 2022 Aug 19. Oncology. 2022. PMID: 35988535
-
Outcome of Patients with Non-Small Cell Lung Cancer and Brain Metastases Treated with Checkpoint Inhibitors.J Thorac Oncol. 2019 Jul;14(7):1244-1254. doi: 10.1016/j.jtho.2019.02.009. Epub 2019 Feb 16. J Thorac Oncol. 2019. PMID: 30780002
-
Management and Outcomes of Newly Diagnosed Non-Small Cell Lung Cancer Patients with Brain Metastases: A Real-World Study in Japan.Oncology. 2020;98(7):460-467. doi: 10.1159/000506368. Epub 2020 Mar 27. Oncology. 2020. PMID: 32222702
-
Therapeutic perspectives for brain metastases in non-oncogene addicted non-small cell lung cancer (NSCLC): Towards a less dismal future?Crit Rev Oncol Hematol. 2018 Aug;128:19-29. doi: 10.1016/j.critrevonc.2018.05.013. Epub 2018 May 24. Crit Rev Oncol Hematol. 2018. PMID: 29958628 Review.
-
Brain Metastases in NSCLC - are TKIs Changing the Treatment Strategy?Anticancer Res. 2015 Nov;35(11):5797-806. Anticancer Res. 2015. PMID: 26504000 Review.
Cited by
-
Prevalence, therapeutic approaches, and survival rates of brain metastases in non-small cell lung cancer: a multi-institutional claims-based study from 2014 to 2024.J Neurooncol. 2025 Oct;175(1):367-377. doi: 10.1007/s11060-025-05139-1. Epub 2025 Jul 21. J Neurooncol. 2025. PMID: 40690182
References
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

