Attenuation of Chronic Inflammation in Intestinal Organoids with Graphene Oxide-Mediated Tumor Necrosis Factor-α_Small Interfering RNA Delivery
- PMID: 38325360
- PMCID: PMC10883062
- DOI: 10.1021/acs.langmuir.3c02741
Attenuation of Chronic Inflammation in Intestinal Organoids with Graphene Oxide-Mediated Tumor Necrosis Factor-α_Small Interfering RNA Delivery
Abstract
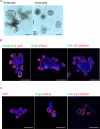
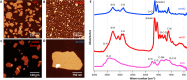
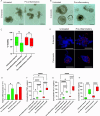
Inflammatory bowel disease (IBD) is a chronic inflammatory disease of the gastrointestinal tract with a complex and multifactorial etiology, making it challenging to treat. While recent advances in immunomodulatory biologics, such as antitumor necrosis factor-α (TNF-α) antibodies, have shown moderate success, systemic administration of antibody therapeutics may lead to several adverse effects, including the risk of autoimmune disorders due to systemic cytokine depletion. Transient RNA interference using exogenous short interfering RNA (siRNA) to regulate target gene expression at the transcript level offers an alternative to systemic immunomodulation. However, siRNAs are susceptible to premature degradation and have poor cellular uptake. Graphene oxide (GO) nanoparticles have been shown to be effective nanocarriers for biologics due to their reduced cytotoxicity and enhanced bioavailability. In this study, we evaluate the therapeutic efficacy of GO mediated TNF-α_siRNA using in vitro models of chronic inflammation generated by treating murine small intestines (enteroids) and large intestines (colonoids) with inflammatory agents IL-1β, TNF-α, and LPS. The organotypic mouse enteroids and colonoids developed an inflammatory phenotype similar to that of IBD, characterized by impaired epithelial homeostasis and an increased production of inflammatory cytokines such as TNF-α, IL-1β, and IL-6. We assessed siRNA delivery to these inflamed organoids using three different GO formulations. Out of the three, small-sized GO with polymer and dendrimer modifications (smGO) demonstrated the highest transfection efficiency, which led to the downregulation of inflammatory cytokines, indicating an attenuation of the inflammatory phenotype. Moreover, the transfection efficiency and inflammation-ameliorating effects could be further enhanced by increasing the TNF-α_siRNA/smGO ratio from 1:1 to 3:1. Overall, the results of this study demonstrate that ex vivo organoids with disease-specific phenotypes are invaluable models for assessing the therapeutic potential of nanocarrier-mediated drug and biologic delivery systems.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
-
Orally delivered thioketal nanoparticles loaded with TNF-α-siRNA target inflammation and inhibit gene expression in the intestines.Nat Mater. 2010 Nov;9(11):923-8. doi: 10.1038/nmat2859. Epub 2010 Oct 10. Nat Mater. 2010. PMID: 20935658 Free PMC article.
-
Optimizing the Intracellular Delivery of Therapeutic Anti-inflammatory TNF-α siRNA to Activated Macrophages Using Lipidoid-Polymer Hybrid Nanoparticles.Front Bioeng Biotechnol. 2021 Jan 14;8:601155. doi: 10.3389/fbioe.2020.601155. eCollection 2020. Front Bioeng Biotechnol. 2021. PMID: 33520957 Free PMC article.
-
Intestinal enteroids/organoids: A novel platform for drug discovery in inflammatory bowel diseases.World J Gastroenterol. 2019 Aug 14;25(30):4125-4147. doi: 10.3748/wjg.v25.i30.4125. World J Gastroenterol. 2019. PMID: 31435168 Free PMC article. Review.
-
γ-Glutamyl cysteine and γ-glutamyl valine inhibit TNF-α signaling in intestinal epithelial cells and reduce inflammation in a mouse model of colitis via allosteric activation of the calcium-sensing receptor.Biochim Biophys Acta. 2015 May;1852(5):792-804. doi: 10.1016/j.bbadis.2014.12.023. Epub 2015 Jan 3. Biochim Biophys Acta. 2015. PMID: 25558818
Cited by
-
Gut Microbial Targets in Inflammatory Bowel Disease: Current Position and Future Developments.Biomedicines. 2025 Mar 14;13(3):716. doi: 10.3390/biomedicines13030716. Biomedicines. 2025. PMID: 40149692 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

