Reversing cancer immunoediting phases with a tumor-activated and optically reinforced immunoscaffold
- PMID: 38333614
- PMCID: PMC10850754
- DOI: 10.1016/j.bioactmat.2024.01.026
Reversing cancer immunoediting phases with a tumor-activated and optically reinforced immunoscaffold
Abstract
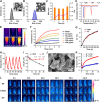
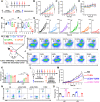
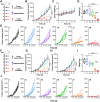
In situ vaccine (ISV) is a promising immunotherapeutic tactic due to its complete tumoral antigenic repertoire. However, its efficiency is limited by extrinsic inevitable immunosuppression and intrinsic immunogenicity scarcity. To break this plight, a tumor-activated and optically reinforced immunoscaffold (TURN) is exploited to trigger cancer immunoediting phases regression, thus levering potent systemic antitumor immune responses. Upon response to tumoral reactive oxygen species, TURN will first release RGX-104 to attenuate excessive immunosuppressive cells and cytokines, and thus immunosuppression falls and immunogenicity rises. Subsequently, intermittent laser irradiation-activated photothermal agents (PL) trigger abundant tumor antigens exposure, which causes immunogenicity springs and preliminary infiltration of T cells. Finally, CD137 agonists from TURN further promotes the proliferation, function, and survival of T cells for durable antitumor effects. Therefore, cancer immunoediting phases reverse and systemic antitumor immune responses occur. TURN achieves over 90 % tumor growth inhibition in both primary and secondary tumor lesions, induces potent systemic immune responses, and triggers superior long-term immune memory in vivo. Taken together, TURN provides a prospective sight for ISV from the perspective of immunoediting phases.
Keywords: Hydrogel; Immunoediting phases; Immunoscaffold; In situ vaccine; Photothermal therapy.
© 2024 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work.
Figures





Similar articles
-
Nanoscale Metal-Organic Frameworks for Cancer Immunotherapy.Acc Chem Res. 2020 Sep 15;53(9):1739-1748. doi: 10.1021/acs.accounts.0c00313. Epub 2020 Aug 18. Acc Chem Res. 2020. PMID: 32808760 Free PMC article.
-
Cancer Immunoediting in the Era of Immuno-oncology.Clin Cancer Res. 2022 Sep 15;28(18):3917-3928. doi: 10.1158/1078-0432.CCR-21-1804. Clin Cancer Res. 2022. PMID: 35594163 Free PMC article.
-
NIR responsive tumor vaccine in situ for photothermal ablation and chemotherapy to trigger robust antitumor immune responses.J Nanobiotechnology. 2021 May 17;19(1):142. doi: 10.1186/s12951-021-00880-x. J Nanobiotechnology. 2021. PMID: 34001148 Free PMC article.
-
In situ vaccination: Harvesting low hanging fruit on the cancer immunotherapy tree.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2019 Jan;11(1):e1524. doi: 10.1002/wnan.1524. Epub 2018 Apr 18. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2019. PMID: 29667346 Review.
-
Cancer immunoediting in malignant glioma.Neurosurgery. 2012 Aug;71(2):201-22; discussion 222-3. doi: 10.1227/NEU.0b013e31824f840d. Neurosurgery. 2012. PMID: 22353795 Review.
Cited by
-
Special issue: Recent advances in immunotherapy and immunoengineering.Bioact Mater. 2025 Feb 28;48:529-530. doi: 10.1016/j.bioactmat.2025.01.038. eCollection 2025 Jun. Bioact Mater. 2025. PMID: 40104023 Free PMC article. No abstract available.
References
LinkOut - more resources
Full Text Sources
Research Materials

