Soluble Angiotensin-Converting Enzyme 2 Protein Improves Survival and Lowers Viral Titers in Lethal Mouse Model of Severe Acute Respiratory Syndrome Coronavirus Type 2 Infection with the Delta Variant
- PMID: 38334597
- PMCID: PMC10854654
- DOI: 10.3390/cells13030203
Soluble Angiotensin-Converting Enzyme 2 Protein Improves Survival and Lowers Viral Titers in Lethal Mouse Model of Severe Acute Respiratory Syndrome Coronavirus Type 2 Infection with the Delta Variant
Abstract
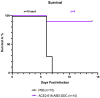
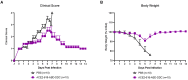
Severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) utilizes angiotensin-converting enzyme 2 (ACE2) as its main receptor for cell entry. We bioengineered a soluble ACE2 protein termed ACE2 618-DDC-ABD that has increased binding to SARS-CoV-2 and prolonged duration of action. Here, we investigated the protective effect of this protein when administered intranasally to k18-hACE2 mice infected with the aggressive SARS-CoV-2 Delta variant. k18-hACE2 mice were infected with the SARS-CoV-2 Delta variant by inoculation of a lethal dose (2 × 104 PFU). ACE2 618-DDC-ABD (10 mg/kg) or PBS was administered intranasally six hours prior and 24 and 48 h post-viral inoculation. All animals in the PBS control group succumbed to the disease on day seven post-infection (0% survival), whereas, in contrast, there was only one casualty in the group that received ACE2 618-DDC-ABD (90% survival). Mice in the ACE2 618-DDC-ABD group had minimal disease as assessed using a clinical score and stable weight, and both brain and lung viral titers were markedly reduced. These findings demonstrate the efficacy of a bioengineered soluble ACE2 decoy with an extended duration of action in protecting against the aggressive Delta SARS-CoV-2 variant. Together with previous work, these findings underline the universal protective potential against current and future emerging SARS-CoV-2 variants.
Keywords: ACE2; COVID-19; SARS-CoV-2; angiotensin-converting enzyme 2.
Conflict of interest statement
D Batlle and J Wysocki are coinventors of patents entitled “Active Low Molecular Weight Variants of Angiotensin Converting Enzyme 2 (ACE2)” and “Soluble ACE2 Variants and Uses therefor”. D Batlle is founder of Angiotensin Therapeutics Inc. D Batlle has received consulting fees from Advicenne unrelated to this work and received unrelated research support from a grant from AstraZeneca. G Randall reports consultancy agreements with Optikira. J Wysocki and J Henkin report scientific advisor capacity for Angiotensin Therapeutics Inc. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

