Time to Anticoagulation Reversal and Outcomes After Intracerebral Hemorrhage
- PMID: 38335064
- PMCID: PMC11002694
- DOI: 10.1001/jamaneurol.2024.0221
Time to Anticoagulation Reversal and Outcomes After Intracerebral Hemorrhage
Abstract
Importance: Intracerebral hemorrhage (ICH) is the deadliest stroke subtype, and mortality rates are especially high in anticoagulation-associated ICH. Recently, specific anticoagulation reversal strategies have been developed, but it is not clear whether there is a time-dependent treatment effect for door-to-treatment (DTT) times in clinical practice.
Objective: To evaluate whether DTT time is associated with outcome among patients with anticoagulation-associated ICH treated with reversal interventions.
Design, setting, and participants: This cohort study used data from the American Heart Association Get With The Guidelines-Stroke quality improvement registry. Patients with ICH who presented within 24 hours of symptom onset across 465 US hospitals from 2015 to 2021 were included. Data were analyzed from January to September 2023.
Exposures: Anticoagulation-associated ICH.
Main outcomes and measures: DTT times and outcomes were analyzed using logistic regression modeling, adjusted for demographic, history, baseline, and hospital characteristics, with hospital-specific random intercepts to account for clustering by site. The primary outcome of interest was the composite inpatient mortality and discharge to hospice. Additional prespecified secondary outcomes, including functional outcome (discharge modified Rankin Scale score, ambulatory status, and discharge venue), were also examined.
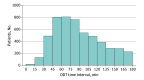
Results: Of 9492 patients with anticoagulation-associated ICH and documented reversal intervention status, 4232 (44.6%) were female, and the median (IQR) age was 77 (68-84) years. A total of 7469 (78.7%) received reversal therapy, including 4616 of 5429 (85.0%) taking warfarin and 2856 of 4069 (70.2%) taking a non-vitamin K antagonist oral anticoagulant. For the 5224 patients taking a reversal intervention with documented workflow times, the median (IQR) onset-to-treatment time was 232 (142-482) minutes and the median (IQR) DTT time was 82 (58-117) minutes, with a DTT time of 60 minutes or less in 1449 (27.7%). A DTT time of 60 minutes or less was associated with decreased mortality and discharge to hospice (adjusted odds ratio, 0.82; 95% CI, 0.69-0.99) but no difference in functional outcome (ie, a modified Rankin Scale score of 0 to 3; adjusted odds ratio, 0.91; 95% CI, 0.67-1.24). Factors associated with a DTT time of 60 minutes or less included White race, higher systolic blood pressure, and lower stroke severity.
Conclusions and relevance: In US hospitals participating in Get With The Guidelines-Stroke, earlier anticoagulation reversal was associated with improved survival for patients with ICH. These findings support intensive efforts to accelerate evaluation and treatment for patients with this devastating form of stroke.
Conflict of interest statement
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources

