Clinical benefit of anti-PD-(L)1 immunotherapies in advanced cancer in France: a population-based estimate from 2014 to 2021
- PMID: 38335904
- PMCID: PMC10937192
- DOI: 10.1016/j.esmoop.2024.102240
Clinical benefit of anti-PD-(L)1 immunotherapies in advanced cancer in France: a population-based estimate from 2014 to 2021
Abstract
Background: In France, the potential benefit of new treatments is initially evaluated by the Haute Autorité de Santé to determine reimbursement and pricing, but rarely afterwards. Although immunotherapies (ITs) have considerably improved the survival of patients, few data are available on their long-term benefit at a population-treated level. The present retrospective study aimed to assess the clinical benefit of ITs compared to the previous standards of care (SoCs) in France from 2014 to 2021.
Materials and methods: To do this, we analyzed all ITs from the anti-programmed cell death protein 1/programmed death-ligand 1 [anti-PD-(L)1] class used in monotherapy or in association with another treatment available in early access or reimbursed in France between 2014 and 2021, regardless of indication. The number of patients initiating an IT was retrieved by year, drug and indication. Using extrapolated Kaplan-Meier curves, utility scores and the population treated, the clinical benefit was expressed as the number of deaths prevented (DP), life-years (LYs) and quality-adjusted life years (QALYs) gained compared to previous SoC.
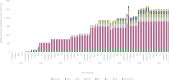
Results: Across the period, five ITs were marketed in 21 indications related to eight primary tumor sites. Between 2014 and 2021, 132 924 patients initiated an IT. By December 2021, 16 173 (13 804-17 141) deaths were delayed compared to previous SoC, mainly in lung cancer. Compared to their SoC, ITs provided a gain of 37 316 (33 581-41 048) additional LYs and 27 709 (23 784-30 450) additional QALYs. Lung cancer was the driver indication with 70.6% of LYs and 68.4% of QALYs gained followed by melanoma with 18.7% and 20.4% of the gain, respectively.
Conclusions: Significant gains in DP, LYs and QALYs have been observed in France following the introduction of ITs.
Keywords: clinical benefit; immunotherapies; life-years; oncology.
Copyright © 2024 Bristol Myers Squibb. Published by Elsevier Ltd.. All rights reserved.
Figures

Similar articles
-
A comparative analysis of two contrasting European approaches for rewarding the value added by drugs for cancer: England versus France.Pharmacoeconomics. 2014 May;32(5):509-20. doi: 10.1007/s40273-014-0144-z. Pharmacoeconomics. 2014. PMID: 24599784
-
Effect of Treatment with the PD-1/PD-L1 Inhibitors on Key Health Outcomes of Cancer Patients.BioDrugs. 2021 Jan;35(1):61-73. doi: 10.1007/s40259-020-00459-2. Epub 2020 Dec 17. BioDrugs. 2021. PMID: 33331991
-
Cost-effectiveness of midostaurin in the treatment of newly diagnosed FLT3-mutated acute myeloid leukemia in France.Eur J Health Econ. 2020 Jun;21(4):543-555. doi: 10.1007/s10198-019-01149-9. Epub 2020 Jan 22. Eur J Health Econ. 2020. PMID: 31970530
-
Comparative survival benefit of currently licensed second or third line treatments for epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK) negative advanced or metastatic non-small cell lung cancer: a systematic review and secondary analysis of trials.BMC Cancer. 2019 Apr 25;19(1):392. doi: 10.1186/s12885-019-5507-6. BMC Cancer. 2019. PMID: 31023244 Free PMC article.
-
Organizational Impact of Immunotherapies in Advanced Cancers in France.JCO Glob Oncol. 2023 Aug;9:e2300026. doi: 10.1200/GO.23.00026. JCO Glob Oncol. 2023. PMID: 37595168 Free PMC article. Review.
Cited by
-
Advances in lymphatic metastasis of non-small cell lung cancer.Cell Commun Signal. 2024 Apr 2;22(1):201. doi: 10.1186/s12964-024-01574-1. Cell Commun Signal. 2024. PMID: 38566083 Free PMC article. Review.
References
-
- Sung H., Ferlay H., Siegel C., et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. - PubMed
-
- Defossez G., Le Guyader-Peyrou S., Uhry Z., et al. Santé Publique France; Saint-Maurice: 2019. Estimations nationales de l’incidence et de la mortalité par cancer en France métropolitaine entre 1990 et 2018. Étude à partir des registres des cancers du réseau Francim. Volume 1 - Tumeurs solides. Compléments par localisation tumorale et sous-sites ou sous-types histologiques.
-
- Direction de la recherche, des études de l’évaluation et des statistiques. L’état de santé de la population en France. Rapport 2017. Principales causes de décès et de morbités; 2017.
-
- Cardoso R., Guo F., Heisser T. Colorectal cancer incidence, mortality, and stage distribution in European countries in the colorectal cancer screening era: an international population-based study. Lancet Oncol. 2021;22(7):1002–1013. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

