A novel nanobody-based immunocytokine of a mutant interleukin-2 as a potential cancer therapeutic
- PMID: 38337114
- PMCID: PMC10857990
- DOI: 10.1186/s13568-023-01648-2
A novel nanobody-based immunocytokine of a mutant interleukin-2 as a potential cancer therapeutic
Abstract
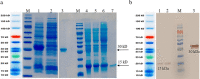
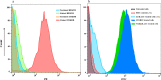
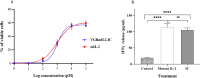
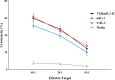
The immunotherapeutic application of interleukin-2 (IL-2) in cancer treatment is limited by its off-target effects on different cell populations and insufficient activation of anti-tumor effector cells at the site of the tumor upon tolerated doses. Targeting IL-2 to the tumor microenvironment by generating antibody-cytokine fusion proteins (immunocytokine) would be a promising approach to increase efficacy without associated toxicity. In this study, a novel nanobody-based immunocytokine is developed by the fusion of a mutant (m) IL-2 with a decreased affinity toward CD25 to an anti-vascular endothelial growth factor receptor-2 (VEGFR2) specific nanobody, denoted as VGRmIL2-IC. The antigen binding, cell proliferation, IFN-γ-secretion, and cytotoxicity of this new immunocytokine are evaluated and compared to mIL-2 alone. Furthermore, the pharmacokinetic properties are analyzed. Flow cytometry analysis shows that the VGRmIL2-IC molecule can selectively target VEGFR2-positive cells. The results reveal that the immunocytokine is comparable to mIL-2 alone in the stimulation of Primary Peripheral Blood Mononuclear Cells (PBMCs) and cytotoxicity in in vitro conditions. In vivo studies demonstrate improved pharmacokinetic properties of VGRmIL2-IC in comparison to the wild or mutant IL-2 proteins. The results presented here suggest VGRmIL2-IC could be considered a candidate for the treatment of VEGFR2-positive tumors.
Keywords: Immunocytokine; Immunotherapy; Mutant IL-2; Nanobody; VEGFR2.
© 2024. The Author(s).
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Melittin-MIL-2 fusion protein as a candidate for cancer immunotherapy.J Transl Med. 2016 Jun 1;14(1):155. doi: 10.1186/s12967-016-0910-0. J Transl Med. 2016. PMID: 27246873 Free PMC article.
-
Cergutuzumab amunaleukin (CEA-IL2v), a CEA-targeted IL-2 variant-based immunocytokine for combination cancer immunotherapy: Overcoming limitations of aldesleukin and conventional IL-2-based immunocytokines.Oncoimmunology. 2017 Jan 11;6(3):e1277306. doi: 10.1080/2162402X.2016.1277306. eCollection 2017. Oncoimmunology. 2017. PMID: 28405498 Free PMC article.
-
Antigen specificity can be irrelevant to immunocytokine efficacy and biodistribution.Proc Natl Acad Sci U S A. 2015 Mar 17;112(11):3320-5. doi: 10.1073/pnas.1416159112. Epub 2015 Mar 2. Proc Natl Acad Sci U S A. 2015. PMID: 25733854 Free PMC article.
-
Anti-cancer Therapies Employing IL-2 Cytokine Tumor Targeting: Contribution of Innate, Adaptive and Immunosuppressive Cells in the Anti-tumor Efficacy.Front Immunol. 2018 Dec 18;9:2905. doi: 10.3389/fimmu.2018.02905. eCollection 2018. Front Immunol. 2018. PMID: 30619269 Free PMC article. Review.
-
Utilizing Immunocytokines for Cancer Therapy.Antibodies (Basel). 2021 Mar 9;10(1):10. doi: 10.3390/antib10010010. Antibodies (Basel). 2021. PMID: 33803078 Free PMC article. Review.
Cited by
-
Efficacy of Anti-Cancer Immune Responses Elicited Using Tumor-Targeted IL-2 Cytokine and Its Derivatives in Combined Preclinical Therapies.Vaccines (Basel). 2025 Jan 13;13(1):69. doi: 10.3390/vaccines13010069. Vaccines (Basel). 2025. PMID: 39852848 Free PMC article. Review.
-
Nanobodies and their derivatives: pioneering the future of cancer immunotherapy.Cell Commun Signal. 2025 Jun 5;23(1):271. doi: 10.1186/s12964-025-02270-4. Cell Commun Signal. 2025. PMID: 40474230 Free PMC article. Review.
References
-
- Atkins MB, Lotze MT, Dutcher JP, Fisher RI, Weiss G, Margolin K, Abrams J, Sznol M, Parkinson D, Hawkins M. High-dose recombinant interleukin 2 therapy for patients with metastatic melanoma: analysis of 270 patients treated between 1985 and 1993. J Clin Oncol. 1999;17(7):2105. doi: 10.1200/JCO.1999.17.7.2105. - DOI - PubMed
-
- Behdani M, Zeinali S, Khanahmad H, Karimipour M, Asadzadeh N, Azadmanesh K, Khabiri A, Schoonooghe S, Anbouhi MH, Hassanzadeh-Ghassabeh G. Generation and characterization of a functional nanobody against the vascular endothelial growth factor receptor-2; angiogenesis cell receptor. Mol Immunol. 2012;50(1–2):35–41. doi: 10.1016/j.molimm.2011.11.013. - DOI - PubMed
-
- Beig Parikhani A, Bagherzadeh K, Dehghan R, Biglari A, Shokrgozar MA, Riazi Rad F, Zeinali S, Talebkhan Y, Ajdary S, Ahangari Cohan R. Human IL-2Rɑ subunit binding modulation of IL-2 through a decline in electrostatic interactions: a computational and experimental approach. PLoS ONE. 2022;17(2):e0264353. doi: 10.1371/journal.pone.0264353. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

