Visuo-Cognitive Phenotypes in Early Multiple Sclerosis: A Multisystem Model of Visual Processing
- PMID: 38337342
- PMCID: PMC10855997
- DOI: 10.3390/jcm13030649
Visuo-Cognitive Phenotypes in Early Multiple Sclerosis: A Multisystem Model of Visual Processing
Abstract
Background: Cognitive impairment can emerge in the earliest stages of multiple sclerosis (MS), with heterogeneity in cognitive deficits often hindering symptom identification and management. Sensory-motor dysfunction, such as visual processing impairment, is also common in early disease and can impact neuropsychological task performance in MS. However, cognitive phenotype research in MS does not currently consider the relationship between early cognitive changes and visual processing impairment.
Objectives: This study explored the relationship between cognition and visual processing in early MS by adopting a three-system model of afferent sensory, central cognitive and efferent ocular motor visual processing to identify distinct visuo-cognitive phenotypes.
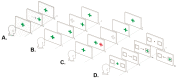
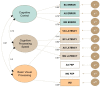
Methods: Patients with clinically isolated syndrome and relapsing-remitting MS underwent neuro-ophthalmic, ocular motor and neuropsychological evaluation to assess each visual processing system. The factor structure of ocular motor variables was examined using exploratory factor analysis, and phenotypes were identified using latent profile analysis.
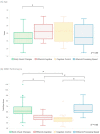
Results: Analyses revealed three ocular-motor constructs (cognitive control, cognitive processing speed and basic visual processing) and four visuo-cognitive phenotypes (early visual changes, efferent-cognitive, cognitive control and afferent-processing speed). While the efferent-cognitive phenotype was present in significantly older patients than was the early visual changes phenotype, there were no other demographic differences between phenotypes. The efferent-cognitive and cognitive control phenotypes had poorer performance on the Symbol Digit Modalities Test compared to that of other phenotypes; however, no other differences in performance were detected.
Conclusion: Our findings suggest that distinct visual processing deficits in early MS may differentially impact cognition, which is not captured using standard neuropsychological evaluation. Further research may facilitate improved symptom identification and intervention in early disease.
Keywords: cognition; multiple sclerosis; phenotypes; visual processing; visuo-cognitive.
Conflict of interest statement
Authors Vagias, Byrne, Millist, Clough and White have no relevant financial or non-financial interests to disclose. Author Fielding received funding from Biogen and Sanofi for research related to this submission. Sanofi was not involved in the design, collection, analysis, interpretation or reporting of the study, but was given the opportunity to review the publication prior to submission. The decision to submit this manuscript for publication was made independently by the authors. Sanofi will be allowed access to all de-identified data from the study for research and audit purposes, if requested.
Figures
References
-
- Cattaneo D., Lamers I., Bertoni R., Feys P., Jonsdottir J. Participation restriction in people with multiple sclerosis: Prevalence and correlations with cognitive, walking, balance, and upper limb impairments. Arch. Phys. Med. Rehabil. 2017;98:1308–1315. doi: 10.1016/j.apmr.2017.02.015. - DOI - PubMed
-
- Hynčicová E., Vyhnálek M., Kalina A., Martinkovič L., Nikolai T., Lisý J., Hort J., Meluzínová E., Laczó J. Cognitive impairment and structural brain changes in patients with clinically isolated syndrome at high risk for multiple sclerosis. J. Neurol. 2017;264:482–493. doi: 10.1007/s00415-016-8368-9. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

