Maternal Vitamin C Intake during Pregnancy Influences Long-Term Offspring Growth with Timing- and Sex-Specific Effects in Guinea Pigs
- PMID: 38337653
- PMCID: PMC10857109
- DOI: 10.3390/nu16030369
Maternal Vitamin C Intake during Pregnancy Influences Long-Term Offspring Growth with Timing- and Sex-Specific Effects in Guinea Pigs
Abstract
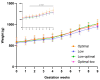
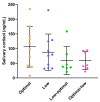
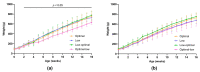
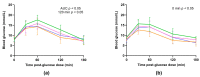
Our previous work in guinea pigs revealed that low vitamin C intake during preconception and pregnancy adversely affects fertility, pregnancy outcomes, and foetal and neonatal growth in a sex-dependent manner. To investigate the long-term impact on offspring, we monitored their growth from birth to adolescence (four months), recorded organ weights at childhood equivalence (28 days) and adolescence, and assessed physiological parameters like oral glucose tolerance and basal cortisol concentrations. We also investigated the effects of the timing of maternal vitamin C restriction (early vs. late gestation) on pregnancy outcomes and the health consequences for offspring. Dunkin Hartley guinea pigs were fed an optimal (900 mg/kg feed) or low (100 mg/kg feed) vitamin C diet ad libitum during preconception. Pregnant dams were then randomised into four feeding regimens: consistently optimal, consistently low, low during early pregnancy, or low during late pregnancy. We found that low maternal vitamin C intake during early pregnancy accelerated foetal and neonatal growth in female offspring and altered glucose homeostasis in the offspring of both sexes at an age equivalent to early childhood. Conversely, low maternal vitamin C intake during late pregnancy resulted in foetal growth restriction and reduced weight gain in male offspring throughout their lifespan. We conclude that altered vitamin C during development has long-lasting, sex-specific consequences for offspring and that the timing of vitamin C depletion is also critical, with low levels during early development being associated with the development of a metabolic syndrome-related phenotype, while later deprivation appears to be linked to a growth-faltering phenotype.
Keywords: ascorbic acid); foetal growth; foetal programming; guinea pig; maternal diet; metabolic function; postnatal growth; pregnancy; vitamin C (ascorbate.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures


Similar articles
-
Effects of Low Vitamin C Intake on Fertility Parameters and Pregnancy Outcomes in Guinea Pigs.Nutrients. 2023 Sep 22;15(19):4107. doi: 10.3390/nu15194107. Nutrients. 2023. PMID: 37836389 Free PMC article.
-
Prolonged maternal vitamin C deficiency overrides preferential fetal ascorbate transport but does not influence perinatal survival in guinea pigs.Br J Nutr. 2013 Nov 14;110(9):1573-9. doi: 10.1017/S0007114513000913. Epub 2013 Apr 16. Br J Nutr. 2013. PMID: 23591139
-
Effect of maternal feed restriction during pregnancy on glucose tolerance in the adult guinea pig.Am J Physiol Regul Integr Comp Physiol. 2003 Jan;284(1):R140-52. doi: 10.1152/ajpregu.00587.2001. Epub 2002 Oct 3. Am J Physiol Regul Integr Comp Physiol. 2003. PMID: 12388450
-
Final report of the safety assessment of L-Ascorbic Acid, Calcium Ascorbate, Magnesium Ascorbate, Magnesium Ascorbyl Phosphate, Sodium Ascorbate, and Sodium Ascorbyl Phosphate as used in cosmetics.Int J Toxicol. 2005;24 Suppl 2:51-111. doi: 10.1080/10915810590953851. Int J Toxicol. 2005. PMID: 16154915 Review.
-
The role of maternal dietary protein on livestock development, production and health.Anim Reprod Sci. 2025 May;276:107835. doi: 10.1016/j.anireprosci.2025.107835. Epub 2025 Apr 1. Anim Reprod Sci. 2025. PMID: 40199102 Review.
References
-
- Smith E.V.L., Dyson R.M., Vanderboor C.M.G., Sarr O., Anderson J., Berry M.J., Regnault T.R.H., Peng L., Gray C. Maternal Fructose Intake Causes Developmental Reprogramming of Hepatic Mitochondrial Catalytic Activity and Lipid Metabolism in Weanling and Young Adult Offspring. Int. J. Mol. Sci. 2022;23:999. doi: 10.3390/ijms23020999. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

