Development of Porcine Accessory Sex Glands
- PMID: 38338105
- PMCID: PMC10854558
- DOI: 10.3390/ani14030462
Development of Porcine Accessory Sex Glands
Abstract
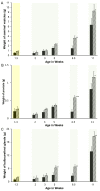
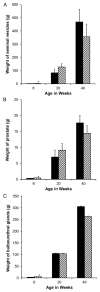
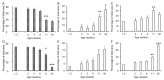
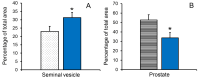
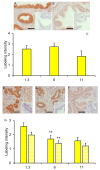
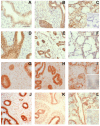
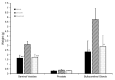
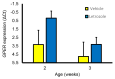
Accessory sex glands are recognized as targets of human disease and may have roles in reproductive success in livestock. The current experiments evaluated the influences of endogenous steroids on the development of porcine accessory sex glands, primarily in the neonatal period. When the aromatase inhibitor, letrozole, was used to inhibit the production of endogenous estrogens in the postnatal interval, growth of the seminal vesicles, prostate, and bulbourethral glands was stimulated. The weights of seminal vesicles, prostate, and bulbourethral glands approximately doubled at 6.5 weeks of age when the reduction in endogenous estrogens began at 1 week of age (p < 0.01). However, by 20 and 40 weeks of age, the weights of accessory sex glands were similar between the letrozole-treated boars and the vehicle-treated littermates indicating the growth stimulation was a transient effect when the treatment interval was short. The presence of both classical nuclear estrogen receptors and the G protein-coupled estrogen receptor in neonatal accessory sex glands indicated multiple signaling pathways might mediate the growth inhibition by endogenous estrogens. The absence of a detectable response when the classical estrogen receptors were blocked with fulvestrant (or when the androgen receptor was blocked with flutamide) suggests that endogenous estrogens act through the G protein-coupled estrogen receptor to inhibit the development of accessory sex glands during this neonatal to early juvenile interval.
Keywords: GPER; bulbourethral glands; estrogen receptor; prostate; seminal vesicles.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Gene Expression in Porcine Bulbourethral Glands.Animals (Basel). 2024 Apr 5;14(7):1115. doi: 10.3390/ani14071115. Animals (Basel). 2024. PMID: 38612354 Free PMC article.
-
Steroid regulation of early postnatal development in the corpus epididymidis of pigs.J Endocrinol. 2015 Jun;225(3):125-34. doi: 10.1530/JOE-15-0001. Epub 2015 Apr 15. J Endocrinol. 2015. PMID: 25876610
-
Reducing endogenous estrogens during the neonatal and juvenile periods affects reproductive tract development and sperm production in postpuberal boars.Anim Reprod Sci. 2008 Dec;109(1-4):218-35. doi: 10.1016/j.anireprosci.2007.10.003. Epub 2007 Oct 13. Anim Reprod Sci. 2008. PMID: 18006255
-
Prolactin influences upon androgen action in male accessory sex organs.Adv Sex Horm Res. 1976;2:425-70. Adv Sex Horm Res. 1976. PMID: 189591 Review.
-
Expression and Role of the G Protein-Coupled Estrogen Receptor (GPR30/GPER) in the Development and Immune Response in Female Reproductive Cancers.Front Endocrinol (Lausanne). 2020 Aug 20;11:544. doi: 10.3389/fendo.2020.00544. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 32973677 Free PMC article. Review.
Cited by
-
Gene Expression in Porcine Bulbourethral Glands.Animals (Basel). 2024 Apr 5;14(7):1115. doi: 10.3390/ani14071115. Animals (Basel). 2024. PMID: 38612354 Free PMC article.
-
The Individual Variations in Sperm Quality of High-Fertility Boars Impact the Offspring Production and Early Physiological Functions.Vet Sci. 2025 Jun 13;12(6):582. doi: 10.3390/vetsci12060582. Vet Sci. 2025. PMID: 40559819 Free PMC article.
References
-
- Di Donato M., Giovannelli P., Cernera G., Di Santi A., Marino I., Bilancio A., Galasso G., Auricchio F., Migliaccio A., Castoria G. Non-genomic androgen action regulates proliferative/migratory signaling in stromal cells. Front. Endocrinol. 2014;5:225. doi: 10.3389/fendo.2014.00225. - DOI - PMC - PubMed
-
- Berger T., McCarthy M., Pearl C.A., At-Taras E., Roser J.F., Conley A. Reducing endogenous estrogens during the neonatal and juvenile periods affects reproductive tract development and sperm production in postpuberal boars. Anim. Reprod. Sci. 2008;109:218–235. doi: 10.1016/j.anireprosci.2007.10.003. - DOI - PubMed

