Experimental In Vitro Microfluidic Calorimetric Chip Data towards the Early Detection of Infection on Implant Surfaces
- PMID: 38339736
- PMCID: PMC10857106
- DOI: 10.3390/s24031019
Experimental In Vitro Microfluidic Calorimetric Chip Data towards the Early Detection of Infection on Implant Surfaces
Abstract
Heat flux measurement shows potential for the early detection of infectious growth. Our research is motivated by the possibility of using heat flux sensors for the early detection of infection on aortic vascular grafts by measuring the onset of bacterial growth. Applying heat flux measurement as an infectious marker on implant surfaces is yet to be experimentally explored. We have previously shown the measurement of the exponential growth curve of a bacterial population in a thermally stabilized laboratory environment. In this work, we further explore the limits of the microcalorimetric measurements via heat flux sensors in a microfluidic chip in a thermally fluctuating environment.
Keywords: heat flux measurement; microbiology; microfluidics.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




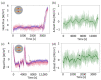
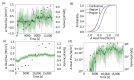
References
-
- Vehusheia S.L.K., Roman C., Braissant O., Arnoldini M., Hierold C. Enabling direct microcalorimetric measurement of metabolic activity and exothermic reactions onto microfluidic platforms via heat flux sensor integration. Microsystems Nanoeng. 2023;9:56. doi: 10.1038/s41378-023-00525-z. - DOI - PMC - PubMed
-
- Hansen L.D., Macfarlane C., McKinnon N., Smith B.N., Criddle R.S. Use of calorespirometric ratios, heat per CO2 and heat per O2, to quantify metabolic paths and energetics of growing cells. Thermochim. Acta. 2004;422:55–61. doi: 10.1016/j.tca.2004.05.033. - DOI
MeSH terms
LinkOut - more resources
Full Text Sources

