C9orf10/Ossa regulates the bone metastasis of established lung adenocarcinoma cell subline H322L-BO4 in a mouse model
- PMID: 38339971
- PMCID: PMC11447824
- DOI: 10.1111/gtc.13103
C9orf10/Ossa regulates the bone metastasis of established lung adenocarcinoma cell subline H322L-BO4 in a mouse model
Abstract
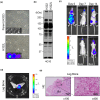
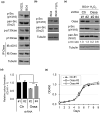
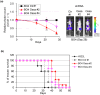
Lung cancer frequently metastasizes to the bones. An in vivo model is urgently required to identify potential therapeutic targets for the prevention and treatment of lung cancer with bone metastasis. We established a lung adenocarcinoma cell subline (H322L-BO4) that specifically showed metastasis to the leg bones and adrenal glands. This was achieved by repeated isolation of metastatic cells from the leg bones of mice. The cells were intracardially injected into nude mice. Survival was prolonged for mice that received H322L-BO4 cells versus original cells (H322L). H322L-BO4 cells did not exhibit obvious changes in general in vitro properties associated with the metastatic potential (e.g., cell growth, migration, and invasion) compared with H322L cells. However, the phosphorylation of chromosome 9 open reading frame 10/oxidative stress-associated Src activator (C9orf10/Ossa) was increased in H322L-BO4 cells. This result confirmed the increased anchorage independence through C9orf10/Ossa-mediated activation of Src family tyrosine kinase. Reduction of C9orf10/Ossa by shRNA reduced cells' metastasis to the leg bone and prolonged survival in mice. These findings indicate that H322L-BO4 cells can be used to evaluate the effect of candidate therapeutic targets against bone metastatic lung cancer cells. Moreover, C9orf10/Ossa may be a useful target for treatment of lung cancer with bone metastasis.
Keywords: C9orf10/Ossa/FAM120A; Src family kinase; anoikis resistance; bone metastasis; cancer metastasis; lung adenocarcinoma; mouse model; signal transduction.
© 2024 The Authors. Genes to Cells published by Molecular Biology Society of Japan and John Wiley & Sons Australia, Ltd.
Figures

References
-
- Annabi, B. , Lee, Y. T. , Turcotte, S. , Naud, E. , Desrosiers, R. R. , Champagne, M. , Eliopoulos, N. , Galipeau, J. , & Béliveau, R. (2003). Hypoxia promotes murine bone‐marrow‐derived stromal cell migration and tube formation. Stem Cells, 21, 337–347. - PubMed
-
- Bartolomé, R. A. , García‐Palmero, I. , Torres, S. , López‐Lucendo, M. , Balyasnikova, I. V. , & Casal, J. I. (2015). IL13 receptor α2 signaling requires a scaffold protein, FAM120A, to activate the FAK and PI3K pathways in colon cancer metastasis. Cancer Research, 75, 2434–2444. - PubMed
-
- Brown, M. T. , & Cooper, J. A. (1996). Regulation, substrates and functions of src. Biochimica et Biophysica Acta, 1287, 121–149. - PubMed
-
- Buijs, J. T. , & van der Pluijm, G. (2009). Osteotropic cancers: From primary tumor to bone. Cancer Letters, 273, 177–193. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

