Cytotoxic leuconoxine-type diazaspiroindole alkaloids isolated from Cryptolepis dubia
- PMID: 38341161
- PMCID: PMC11034800
- DOI: 10.1016/j.bmcl.2024.129650
Cytotoxic leuconoxine-type diazaspiroindole alkaloids isolated from Cryptolepis dubia
Abstract
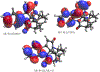
Two leuconoxine-type diazaspiroindole alkaloids, the known compound, (+)-melodinine E (1), and its new analogue, (+)-11-chloromelodinine E (2), were isolated from the stems of Cryptolepis dubia (Burm.f.) M.R. Almeida (Apocynaceae), collected in Laos. The chemical structures of these compounds were determined by analysis of their spectroscopic data and by comparison of these data with literature values, of which the molecular structure of 1 has been determined previously by analysis of its single-crystal X-ray diffraction data. The absolute configurations of 1 and 2 have been defined by their experimental and simulated electronic circular dichroism (ECD) spectroscopic data and supported by 1H and 13C NMR-based DP4+ probability analysis and specific rotation calculations. When tested against a small panel of human cancer cell lines, these two compounds exhibited selective cytotoxicity toward OVCAR3 human ovarian cancer cells.
Keywords: Absolute configuration; Cryptolepis dubia; Leuconoxine-type diazaspiroindole alkaloids; Melodinine E; Selective cytotoxicity.
Copyright © 2024 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
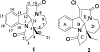
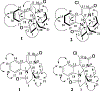
 , 1H
, 1H  1H) and key HMBC (
1H) and key HMBC ( , 1H
, 1H  13C) (upper) and selected NOESY (
13C) (upper) and selected NOESY ( , 1H
, 1H  1H) (lower) correlations for 1 and 2.
1H) (lower) correlations for 1 and 2.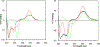
Similar articles
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Topotecan, pegylated liposomal doxorubicin hydrochloride and paclitaxel for second-line or subsequent treatment of advanced ovarian cancer: a systematic review and economic evaluation.Health Technol Assess. 2006 Mar;10(9):1-132. iii-iv. doi: 10.3310/hta10090. Health Technol Assess. 2006. PMID: 16545208
-
Mavacurane-aspidospermane bisindole alkaloids from Hunteria zeylanica: structural elucidation and revision, bioinspired semisynthesis and cytotoxicity.Phytochemistry. 2025 Oct;238:114550. doi: 10.1016/j.phytochem.2025.114550. Epub 2025 May 19. Phytochemistry. 2025. PMID: 40398525
-
An update review on monoterpene indole alkaloids and biological activities of Tabernaemontana species occurring in Brazil.J Ethnopharmacol. 2024 Jun 28;328:117921. doi: 10.1016/j.jep.2024.117921. Epub 2024 Feb 16. J Ethnopharmacol. 2024. PMID: 38369065 Review.
-
The Cytotoxic Cardiac Glycoside (-)-Cryptanoside A from the Stems of Cryptolepis dubia and Its Molecular Targets.J Nat Prod. 2023 Jun 23;86(6):1411-1419. doi: 10.1021/acs.jnatprod.3c00094. Epub 2023 May 22. J Nat Prod. 2023. PMID: 37216676 Free PMC article.
Cited by
-
Concise Total Syntheses of Leuconoxine-Type Alkaloids Enabled by Palladium/Norbornene-Catalyzed Pyrrole Difunctionalization.Angew Chem Int Ed Engl. 2025 Jun 2;64(23):e202502736. doi: 10.1002/anie.202502736. Epub 2025 Apr 10. Angew Chem Int Ed Engl. 2025. PMID: 40163013 Free PMC article.
References
-
- Hanprasertpong N, Teekachunhatean S, Chaiwongsa R, Ongchai S, Kunanusorn P, Sangdee C, Panthong A, Bunteang S, Nathasaen N, Reutrakul V. Analgesic, anti-inflammatory, and chondroprotective activities of Cryptolepis buchananii extract: In vitro and in vivo studies. BioMed Res. Int. 2014;978582. DOI: 10.1155/2014/978582. - DOI - PMC - PubMed
-
- Priyanka Bisht AS, Juyal D. Antimicrobial potential of root part of Cryptolepis buchanani (Roem. and Schult). Pharm Innov J. 2018;7:39–41.
-
- Vasanth S, Gopal RH, Rao RB. Antibacterial activity of Cryptolepis buchananii. Fitoterapia 1997;68:463–464.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

