Randomized trial of anakinra plus zinc vs. prednisone for severe alcohol-associated hepatitis
- PMID: 38342441
- PMCID: PMC11214682
- DOI: 10.1016/j.jhep.2024.01.031
Randomized trial of anakinra plus zinc vs. prednisone for severe alcohol-associated hepatitis
Abstract
Background & aims: Severe alcohol-associated hepatitis (SAH) is associated with high 90-day mortality. Glucocorticoid therapy for 28 days improves 30- but not 90-day survival. We assessed the efficacy and safety of a combination of anakinra, an IL-1 antagonist, plus zinc (A+Z) compared to prednisone using the Day-7 Lille score as a stopping rule in patients with SAH.
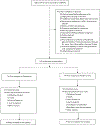
Methods: In this phase IIb double-blind randomized trial in adults with SAH and MELD scores of 20-35, participants were randomized to receive either daily anakinra 100 mg subcutaneously for 14 days plus daily zinc sulfate 220 mg orally for 90 days, or daily prednisone 40 mg orally for 30 days. Prednisone or prednisone placebo was stopped if Day-7 Lille score was >0.45. All study drugs were stopped for uncontrolled infection or ≥5 point increase in MELD score. The primary endpoint was overall survival at 90 days.
Results: Seventy-three participants were randomized to prednisone and 74 to A+Z. The trial was stopped early after a prespecified interim analysis showed prednisone was associated with higher 90-day overall survival (90% vs. 70%; hazard ratio for death = 0.34, 95% CI 0.14-0.83, p = 0.018) and transplant-free survival (88% vs. 64%; hazard ratio for transplant or death = 0.30, 95% CI 0.13-0.69, p = 0.004) than A+Z. Acute kidney injury was more frequent with A+Z (45%) than prednisone (22%) (p = 0.001), but rates of infection were similar (31% in A+Z vs. 27% in prednisone, p = 0.389).
Conclusions: Participants with SAH treated with prednisone using the Day-7 Lille score as a stopping rule had significantly higher overall and transplant-free 90-day survival and lower incidence of acute kidney injury than those treated with A+Z.
Impact and implications: There is no approved treatment for severe alcohol-associated hepatitis (SAH). In this double-blind randomized trial, patients with SAH treated with prednisone using the Lille stopping rule on Day 7 had higher 90-day overall and transplant-free survival and lower rates of acute kidney injury compared to patients treated with a combination of anakinra and zinc. The data support continued use of glucocorticoids for patients with SAH, with treatment discontinuation for those with a Lille score >0.45 on Day 7.
Trial registration: NCT04072822.
Keywords: Corticosteroids; IL1-inhibitor; alcoholic hepatitis; anakinra; survival.
Copyright © 2024 European Association for the Study of the Liver. All rights reserved.
Figures
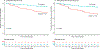



References
-
- Morales-Arráez D, Ventura-Cots M, Altamirano J, et al. The MELD Score Is Superior to the Maddrey Discriminant Function Score to Predict Short-Term Mortality in Alcohol-Associated Hepatitis: A Global Study. Am J Gastroenterol. Feb 1 2022;117(2):301–310. doi: 10.14309/ajg.0000000000001596 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

